QUESTION IMAGE
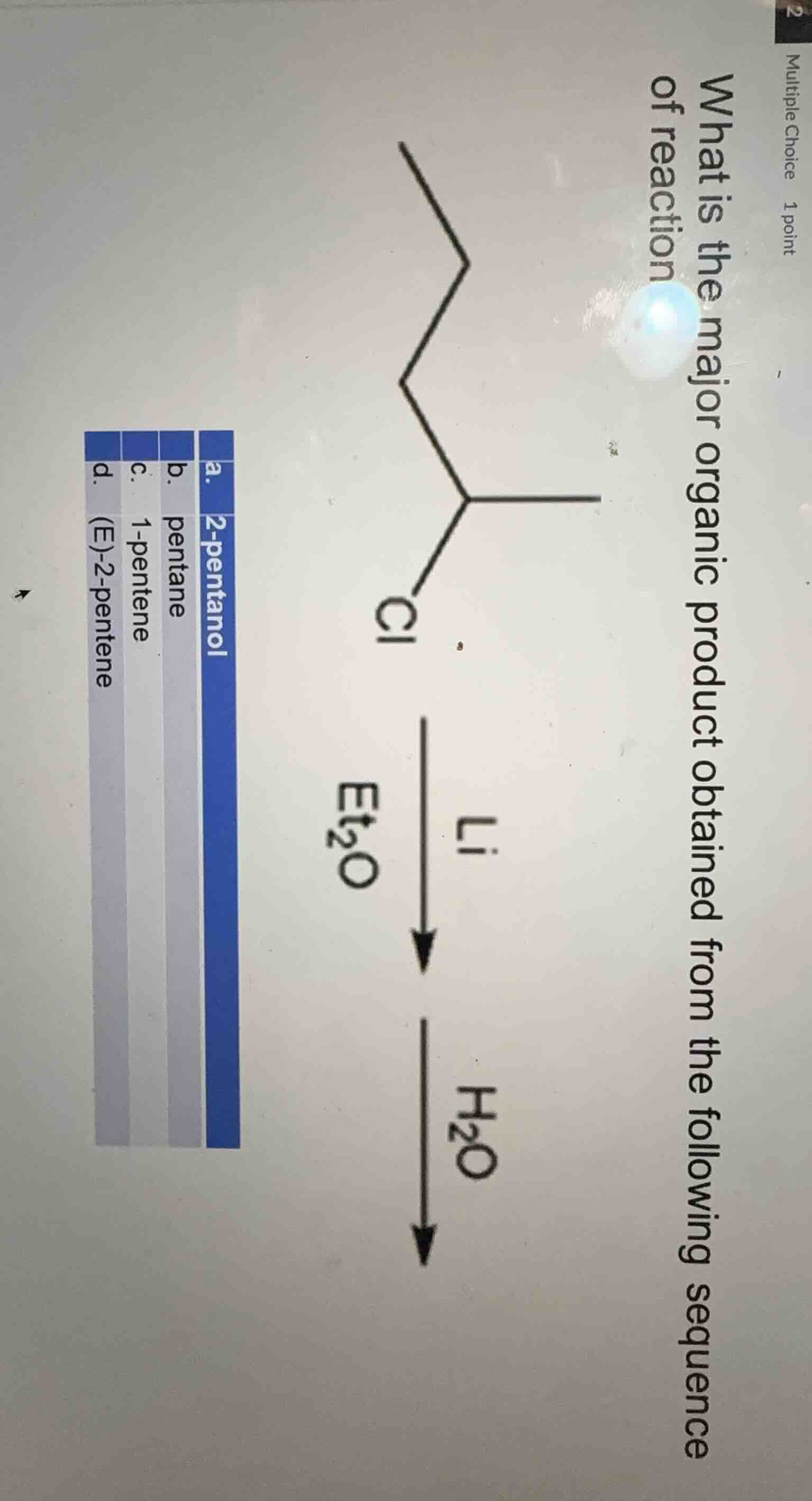
Question
what is the major organic product obtained from the following sequence of reaction
a. 2 - pentanol
b. pentane
c. 1 - pentene
d. (e) - 2 - pentene
Brief Explanations
- First, the reaction of the alkyl chloride (2 - chloropentane) with Li in Et₂O is a formation of an organolithium compound, but then the subsequent reaction with H₂O is a protonation (hydrolysis) to replace the -Cl (after the organolithium step, but actually, the key here is the reduction: Li in Et₂O followed by H₂O is a way to reduce alkyl halides to alkanes. The alkyl chloride here is 2 - chloropentane, and reduction with Li (similar to a radical or ionic reduction) followed by protonation gives pentane. Let's analyze the options:
- Option a: 2 - pentanol would be formed if there was a hydrolysis of an alcohol - forming reaction, but here we have an alkyl halide with Li and then H₂O, not a reaction to form an alcohol.
- Option b: Pentane. The reaction of alkyl halide (2 - chloropentane) with Li (in Et₂O, a common solvent for organolithium reactions, but here the overall process with H₂O is a reduction of the alkyl halide to the alkane. The mechanism: Li donates an electron to the alkyl chloride, forming a radical - anion, then further reduction and protonation from H₂O gives the alkane (pentane, since the parent chain is pentane, and the Cl is on the 2nd carbon, but reduction removes the Cl and replaces it with H, giving pentane).
- Option c: 1 - pentene would be a product of elimination, but the reagents Li/Et₂O and then H₂O are for reduction, not elimination. Elimination would require strong bases or other conditions, not this sequence.
- Option d: (E) - 2 - pentene is also an elimination product, and the given reagents don't promote elimination; they promote reduction of the alkyl halide to alkane.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b. pentane