QUESTION IMAGE
Question
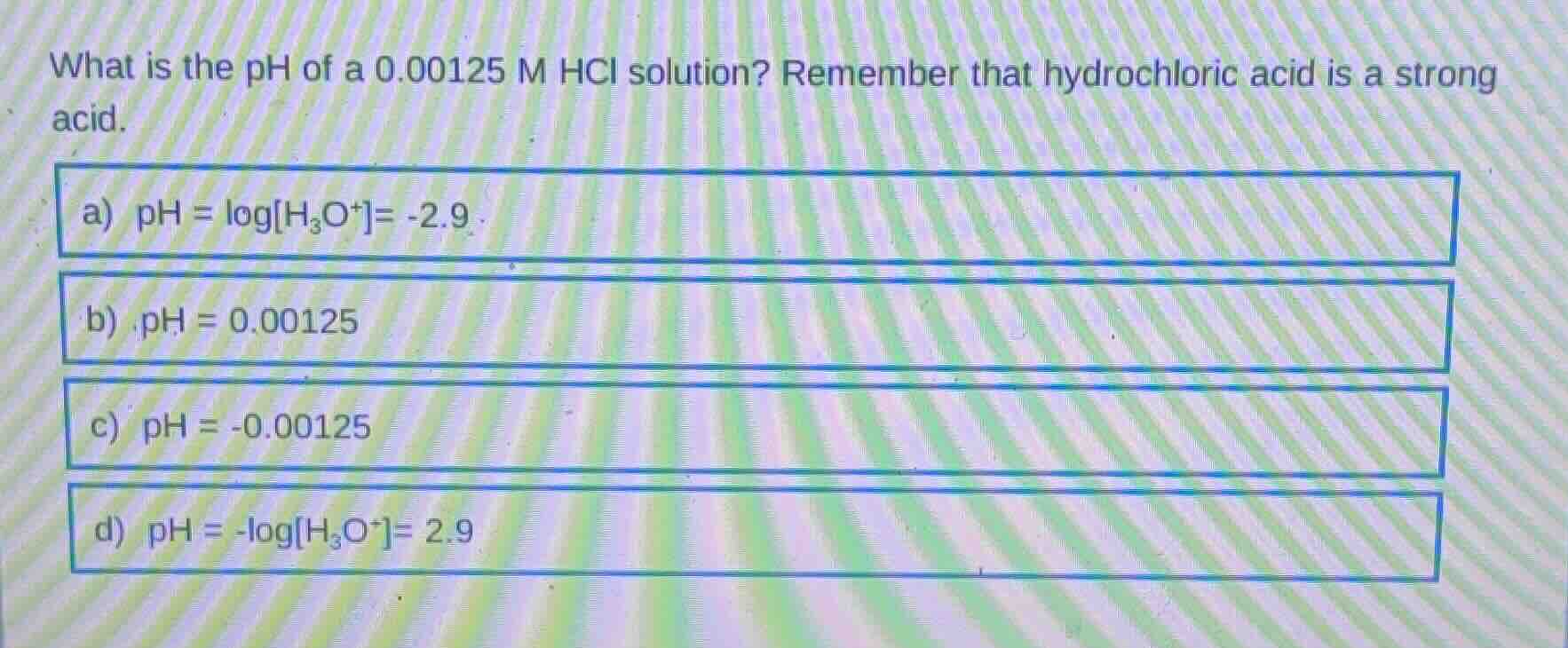
what is the ph of a 0.00125 m hcl solution? remember that hydrochloric acid is a strong acid. a) ph = logh₃o⁺= -2.9 b) ph = 0.00125 c) ph = -0.00125 d) ph = -logh₃o⁺= 2.9
Step1: Find $[H_3O^+]$ concentration
Since HCl is a strong acid, it dissociates completely: $[H_3O^+] = 0.00125\ \text{M}$
Step2: Apply pH formula
pH is defined as $pH = -\log[H_3O^+]$
Substitute value: $pH = -\log(0.00125)$
Calculate: $pH = 2.9$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d) pH = -log[H₃O⁺]= 2.9