QUESTION IMAGE
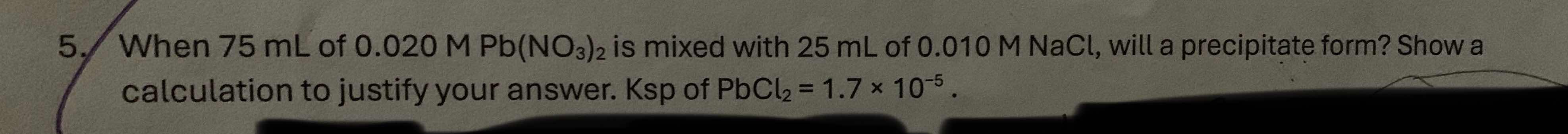
Question
- when 75 ml of 0.020 m $\text{pb(no}_3\text{)}_2$ is mixed with 25 ml of 0.010 m nacl, will a precipitate form? show a calculation to justify your answer. ksp of $\text{pbcl}_2 = 1.7 \times 10^{-5}$.
Step1: Calculate total solution volume
$V_{total} = 75\ \text{mL} + 25\ \text{mL} = 100\ \text{mL} = 0.100\ \text{L}$
Step2: Find $[\text{Pb}^{2+}]$ after dilution
Moles of $\text{Pb}^{2+} = 0.020\ \text{M} \times 0.075\ \text{L} = 0.0015\ \text{mol}$
$[\text{Pb}^{2+}] = \frac{0.0015\ \text{mol}}{0.100\ \text{L}} = 0.015\ \text{M}$
Step3: Find $[\text{Cl}^-]$ after dilution
Moles of $\text{Cl}^- = 0.010\ \text{M} \times 0.025\ \text{L} = 0.00025\ \text{mol}$
$[\text{Cl}^-] = \frac{0.00025\ \text{mol}}{0.100\ \text{L}} = 0.0025\ \text{M}$
Step4: Calculate ion product $Q$
For $\text{PbCl}_2$, $Q = [\text{Pb}^{2+}][\text{Cl}^-]^2$
$Q = 0.015 \times (0.0025)^2 = 0.015 \times 6.25 \times 10^{-6} = 9.375 \times 10^{-8}$
Step5: Compare $Q$ to $K_{sp}$
$K_{sp} = 1.7 \times 10^{-5}$, and $9.375 \times 10^{-8} < 1.7 \times 10^{-5}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Since the ion product $Q = 9.375 \times 10^{-8}$ is less than the solubility product constant $K_{sp} = 1.7 \times 10^{-5}$, no precipitate will form.