QUESTION IMAGE
Question
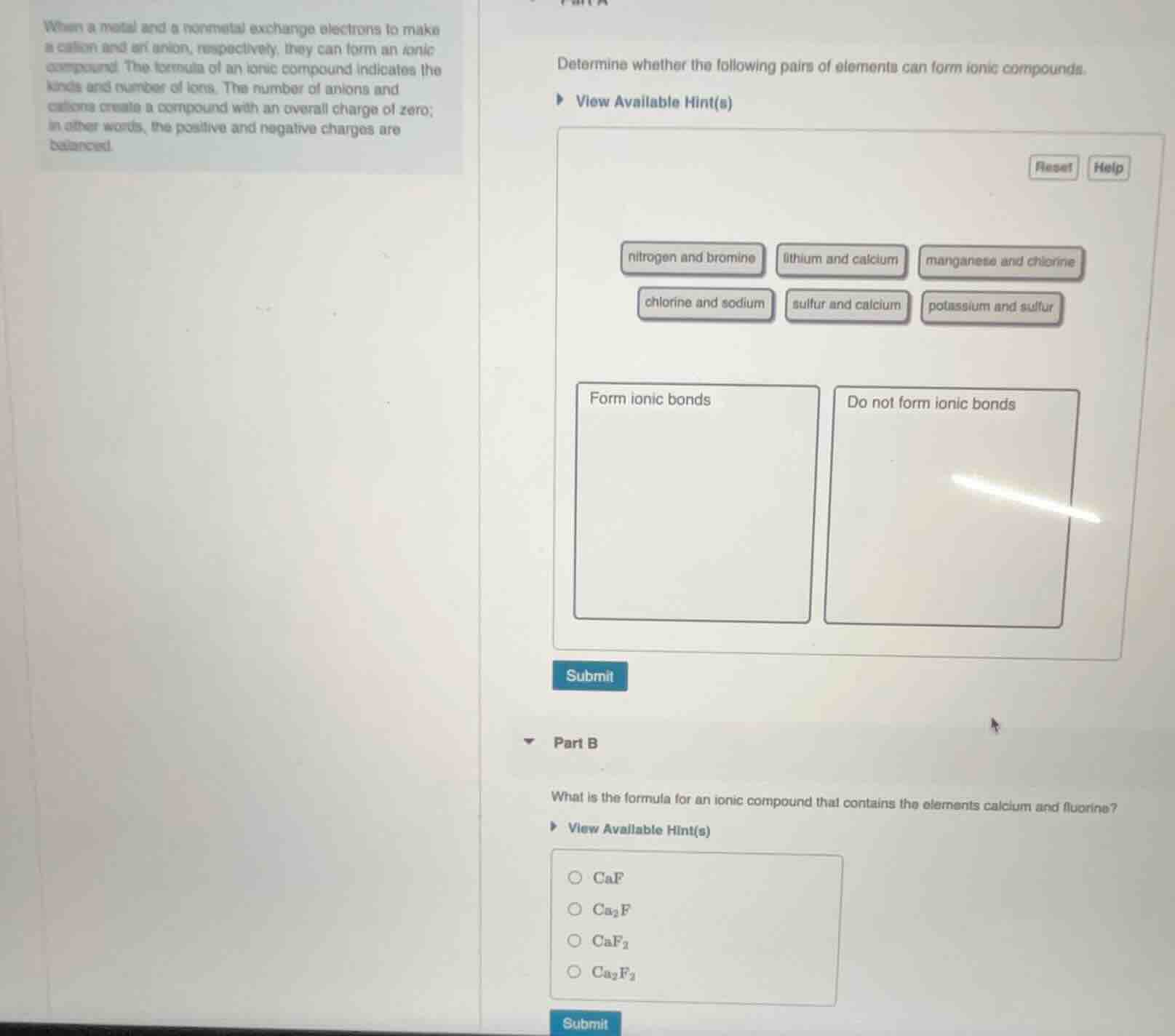
when a metal and a nonmetal exchange electrons to make a cation and an anion, respectively, they can form an ionic compound. the formula of an ionic compound indicates the kinds and number of ions. the number of anions and cations create a compound with an overall charge of zero; in other words, the positive and negative charges are balanced.
part a
determine whether the following pairs of elements can form ionic compounds.
view available hint(s)
reset help
nitrogen and bromine lithium and calcium manganese and chlorine
chlorine and sodium sulfur and calcium potassium and sulfur
form ionic bonds do not form ionic bonds
submit
part b
what is the formula for an ionic compound that contains the elements calcium and fluorine?
view available hint(s)
caf
ca₂f
caf₂
ca₂f₂
submit
Part A
To determine if elements form ionic compounds, we check if one is a metal and the other a non - metal (ionic bonds form between metals and non - metals).
- Nitrogen (non - metal) and bromine (non - metal): Both are non - metals, so they do not form ionic bonds.
- Lithium (metal) and calcium (metal): Both are metals, so they do not form ionic bonds.
- Manganese (metal) and chlorine (non - metal): Metal and non - metal, so they form ionic bonds.
- Chlorine (non - metal) and sodium (metal): Metal and non - metal, so they form ionic bonds.
- Sulfur (non - metal) and calcium (metal): Metal and non - metal, so they form ionic bonds.
- Potassium (metal) and sulfur (non - metal): Metal and non - metal, so they form ionic bonds.
Step 1: Determine the charges of the ions
Calcium (Ca) is in group 2 of the periodic table, so it forms a \(Ca^{2+}\) ion (loses 2 electrons). Fluorine (F) is in group 17, so it forms a \(F^-\) ion (gains 1 electron).
Step 2: Balance the charges
To balance the positive and negative charges, we need 2 fluoride ions (\(F^-\)) for every 1 calcium ion (\(Ca^{2+}\)) because the charge of \(Ca^{2+}\) is +2 and the charge of each \(F^-\) is - 1. The formula is determined by the ratio of ions that gives a net charge of zero. So, the formula is \(CaF_2\) (since \(2\times( - 1)+( + 2)=0\)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Form ionic bonds: manganese and chlorine, chlorine and sodium, sulfur and calcium, potassium and sulfur
Do not form ionic bonds: nitrogen and bromine, lithium and calcium