QUESTION IMAGE
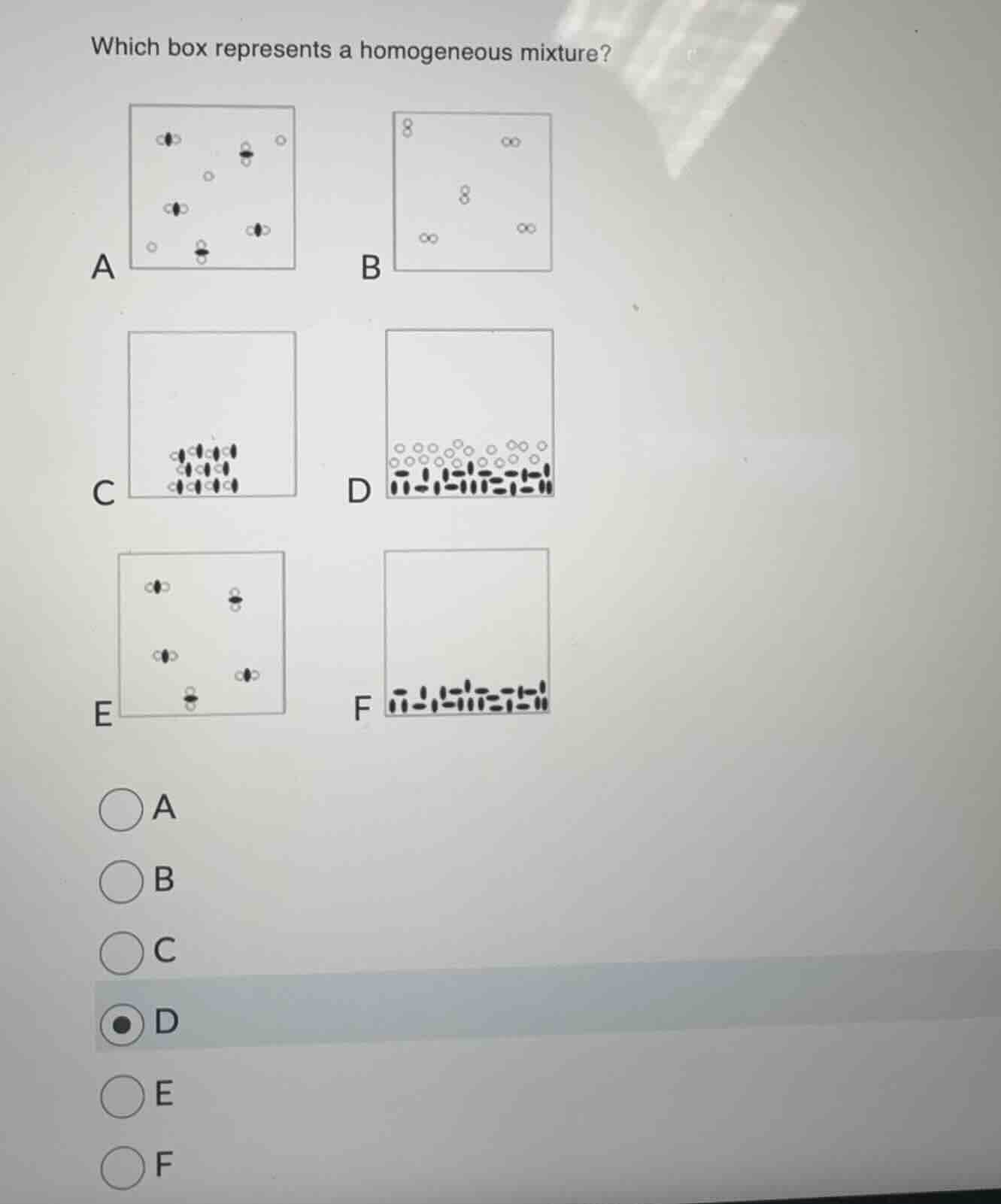
Question
which box represents a homogeneous mixture?
a b
c d
e f
a
b
c
d
e
f
Brief Explanations
A homogeneous mixture has a uniform composition throughout. Let's analyze each box:
- Box A: Contains different types of particles (some with black centers, some white single circles) and is not uniform.
- Box B: Contains only one type of molecule (all the same "8 - shaped" molecules), so it's a pure substance, not a mixture.
- Box C: Contains a single type of structured particle, so it's a pure substance (maybe a compound or element in a lattice), not a mixture.
- Box D: Wait, no—wait, actually, let's re - evaluate. Wait, a homogeneous mixture should have different substances mixed uniformly. Wait, maybe I made a mistake earlier. Wait, let's check again:
- Box A: Has two types of particles (the ones with black in the middle and the white single circles) mixed randomly? No, wait, no—wait, the correct approach: A homogeneous mixture is a mixture with uniform composition (like a solution). A pure substance has only one type of particle (element or compound).
- Box B: All particles are the same (same molecule), so pure substance (element or compound).
- Box C: All particles are the same (structured), pure substance.
- Box D: Has two types of particles (the white circles and the black - lined particles) but they are separated (layers), so that's a heterogeneous mixture (like a mixture of two immiscible substances, layers).
- Box E: Wait, no—wait, let's look at Box A again. Wait, maybe I messed up. Wait, the key is: A homogeneous mixture has different substances (different particles) distributed uniformly. Let's check each:
- Box A: Contains two types of particles (the molecules with black in the middle and the single white circles) and they are mixed throughout the box—so uniform distribution of different particles. Wait, but maybe the molecules with black in the middle are one substance and the single white circles are another. So that would be a homogeneous mixture. But wait, the original selection was D, which is wrong. Wait, no—maybe the particles: Let's think about elements and compounds. A molecule with two atoms (like Box B: all same molecules, so pure substance, maybe an element like O₂). Box A: has two types of particles—one is a molecule with a black atom (maybe a compound like H₂O with a black atom, and single white atoms (maybe H or O)). Wait, no—maybe the correct answer is A? Wait, no, let's start over.
- Define Homogeneous Mixture: A mixture where the components are uniformly distributed (you can't see distinct phases, and the composition is the same throughout). A pure substance has only one type of particle (element: same atom; compound: same molecule). A heterogeneous mixture has non - uniform composition (like layers, clumps).
- Analyze each box:
- Box A: Contains two types of particles (the molecules with a black center and the single white circles) and they are spread out throughout the box—uniform distribution. So this is a homogeneous mixture (different substances mixed uniformly).
- Box B: All particles are the same (same molecule), so pure substance (not a mixture).
- Box C: All particles are the same (structured), pure substance (not a mixture).
- Box D: Two types of particles (white circles and black - lined particles) are in layers (separated), so heterogeneous mixture (non - uniform).
- Box E: Contains two types of molecules (some with black in the center, some with white in the center) mixed uniformly? Wait, Box E has two types of molecules (the ones with black in the middle and the ones wit…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Box A