QUESTION IMAGE
Question
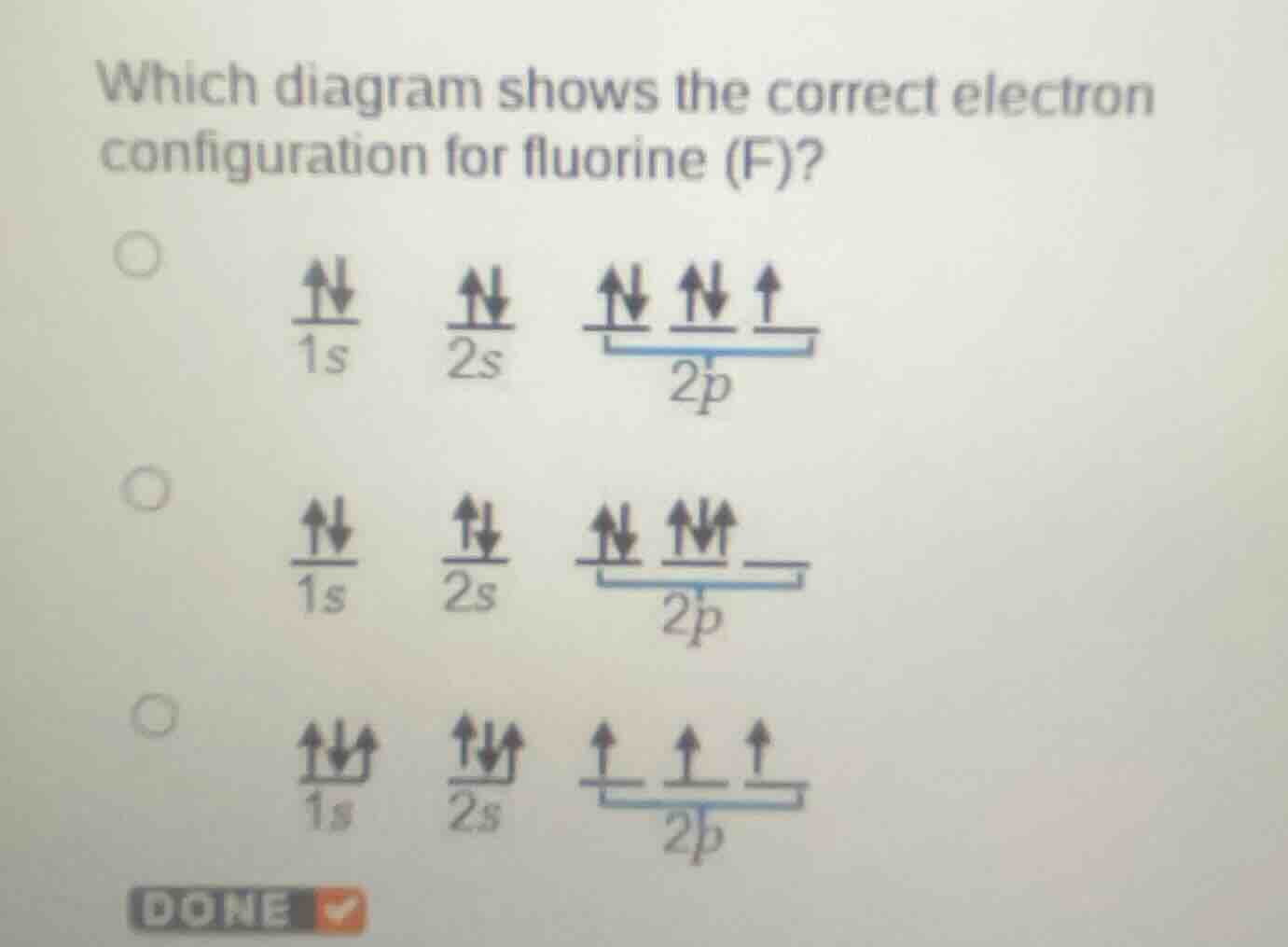
which diagram shows the correct electron configuration for fluorine (f)?
Brief Explanations
- First, recall the electron configuration of fluorine (F). The atomic number of F is 9, so it has 9 electrons.
- The electron configuration follows the Aufbau principle, Pauli exclusion principle, and Hund's rule.
- The \(1s\) orbital can hold 2 electrons (paired, \(\uparrow\downarrow\)).
- The \(2s\) orbital can hold 2 electrons (paired, \(\uparrow\downarrow\)).
- The \(2p\) orbital has 3 sub - orbitals. For 5 electrons in \(2p\) (since \(1s^22s^22p^5\)), according to Hund's rule, electrons fill orbitals singly first with parallel spins, then pair up. So in \(2p\), we have two paired electrons (in two of the \(2p\) sub - orbitals) and one unpaired electron (in the third \(2p\) sub - orbital).
- Looking at the options:
- The first option has \(1s\) (\(\uparrow\downarrow\)), \(2s\) (\(\uparrow\downarrow\)), and \(2p\) with two paired (\(\uparrow\downarrow\)) and one unpaired (\(\uparrow\)) electrons, which matches the electron configuration of F (\(1s^22s^22p^5\)).
- The second option violates Hund's rule as electrons are paired in a \(2p\) sub - orbital before all sub - orbitals have one electron.
- The third option violates the Pauli exclusion principle (more than 2 electrons in \(1s\) and \(2s\) orbitals) and also the correct filling order.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The first option (with \(1s\) \(\uparrow\downarrow\), \(2s\) \(\uparrow\downarrow\), and \(2p\) with two \(\uparrow\downarrow\) and one \(\uparrow\) electrons)