QUESTION IMAGE
Question
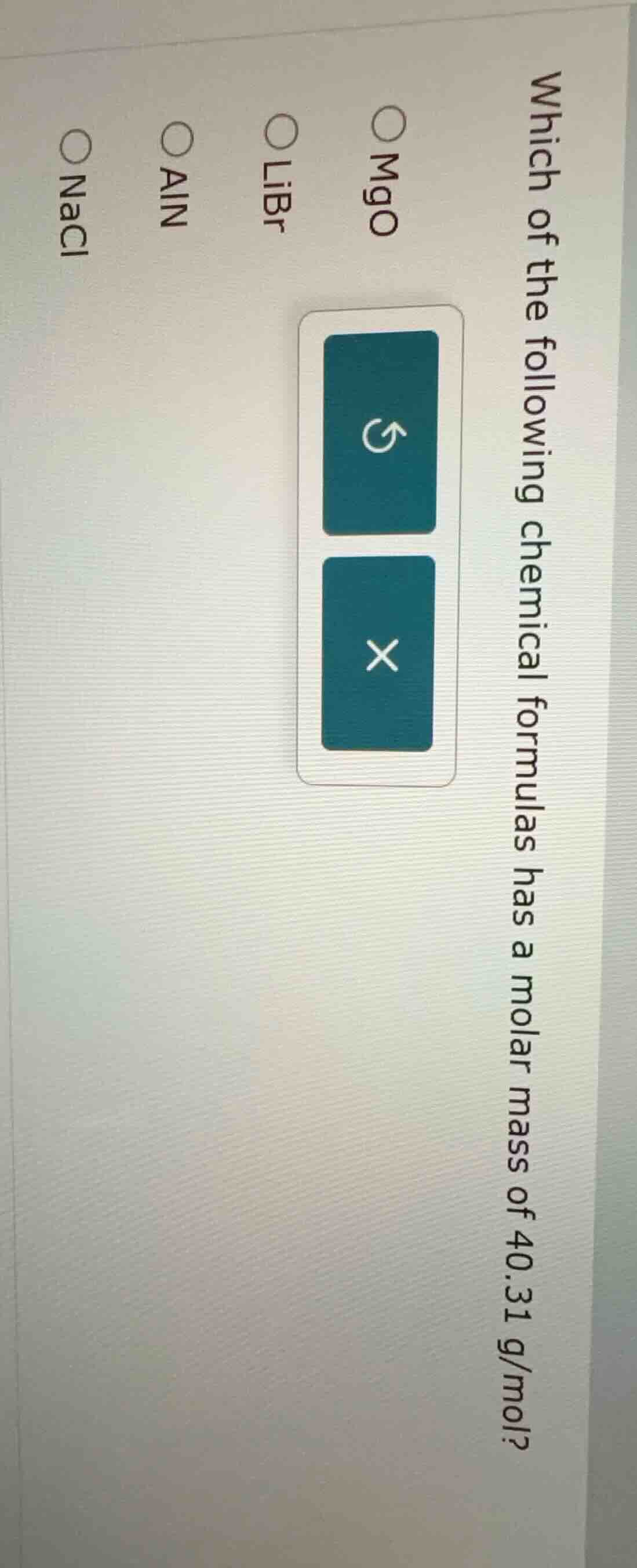
which of the following chemical formulas has a molar mass of 40.31 g/mol?
○ mgo
○ libr
○ aln
○ nacl
Step1: Calculate molar mass of MgO
Molar mass of Mg: $24.31\ g/mol$, molar mass of O: $16.00\ g/mol$.
Molar mass of MgO = $24.31 + 16.00 = 40.31\ g/mol$.
Step2: Calculate molar mass of LiBr
Molar mass of Li: $6.94\ g/mol$, molar mass of Br: $79.90\ g/mol$.
Molar mass of LiBr = $6.94 + 79.90 = 86.84\ g/mol$.
Step3: Calculate molar mass of AlN
Molar mass of Al: $26.98\ g/mol$, molar mass of N: $14.01\ g/mol$.
Molar mass of AlN = $26.98 + 14.01 = 40.99\ g/mol$.
Step4: Calculate molar mass of NaCl
Molar mass of Na: $22.99\ g/mol$, molar mass of Cl: $35.45\ g/mol$.
Molar mass of NaCl = $22.99 + 35.45 = 58.44\ g/mol$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
MgO (the chemical formula with molar mass 40.31 g/mol is MgO)