QUESTION IMAGE
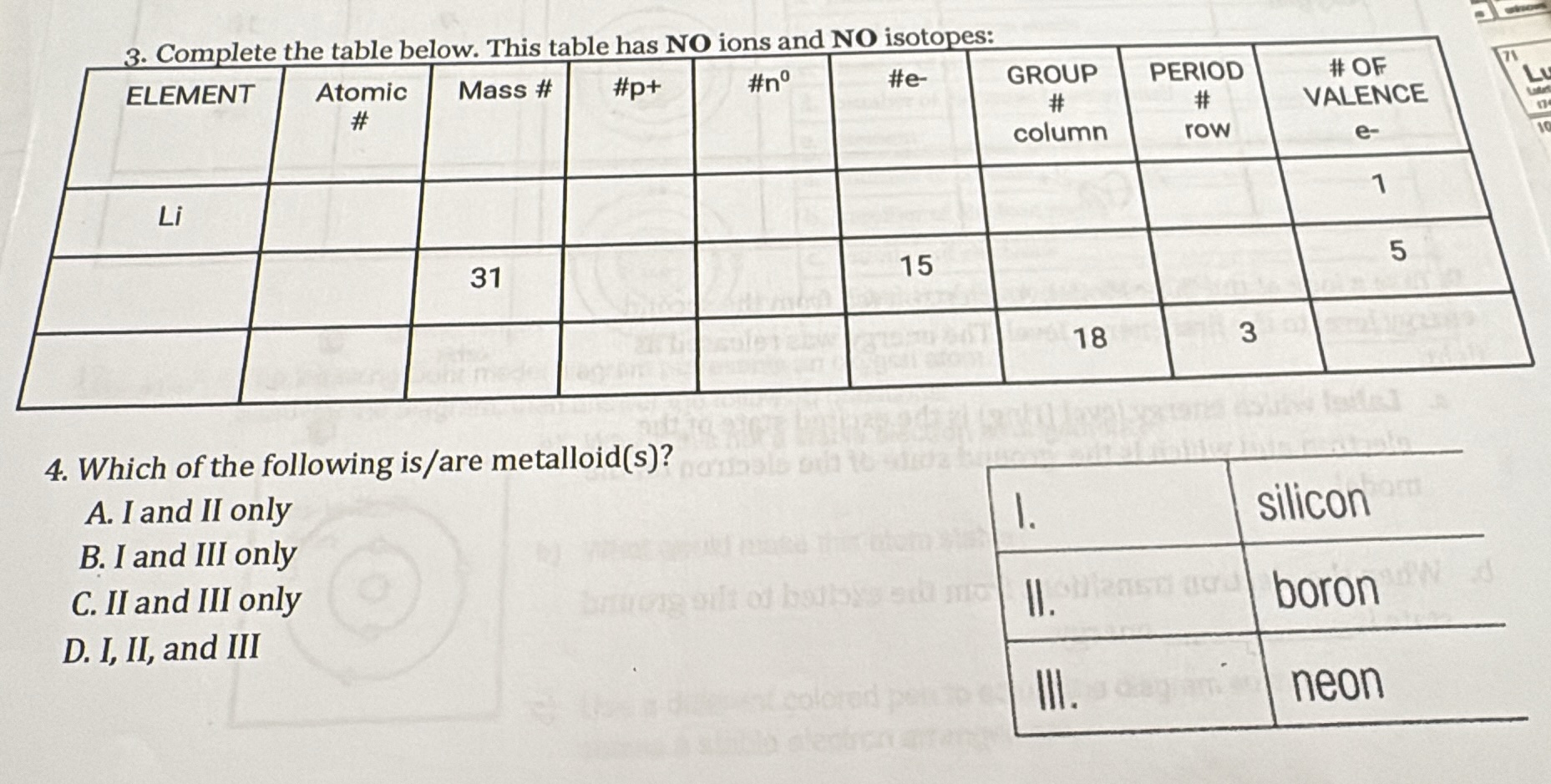
Question
- which of the following is/are metalloid(s)?
a. i and ii only
b. i and iii only
c. ii and iii only
d. i, ii, and iii
i. silicon
ii. boron
iii. neon
Response
Question 3: Completing the Table
To solve this, we use the following relationships:
- Atomic # = #p⁺ = #e⁻ (for neutral atoms)
- Mass # = #p⁺ + #n⁰
- Group # relates to valence electrons (e.g., Group 1 has 1 valence e⁻, Group 15 has 5, Group 18 has 8).
- Period # is the row number (Li is in Period 2, etc.).
Row 1: Li (Lithium)
- Atomic # = 3 (Li’s atomic number is 3)
- #p⁺ = 3 (atomic # = #p⁺)
- #e⁻ = 3 (neutral, so #e⁻ = #p⁺)
- Mass # = 3 (p⁺) + 4 (n⁰, typical Li isotope) = 7
- Group # = 1 (valence e⁻ = 1)
- Period # = 2 (Li is in Period 2)
Row 2: Element with #e⁻ = 15 (neutral, so #p⁺ = 15)
- Atomic # = 15 (Phosphorus, P)
- #p⁺ = 15
- Mass # = 31 (given), so #n⁰ = 31 - 15 = 16
- Group # = 15 (valence e⁻ = 5)
- Period # = 3 (P is in Period 3)
Row 3: Group 18, Period 3
- Group 18 elements have 8 valence e⁻.
- Period 3, Group 18: Argon (Ar)
- Atomic # = 18 (Ar’s atomic number)
- #p⁺ = 18
- #e⁻ = 18 (neutral)
- Mass # = 18 (p⁺) + 22 (n⁰, typical Ar isotope) = 40 (or 39/40; we use 40 for simplicity)
- #n⁰ = 40 - 18 = 22
Filled Table:
| ELEMENT | Atomic # | Mass # | #p⁺ | #n⁰ | #e⁻ | GROUP # | PERIOD # | # OF VALENCE e⁻ |
|---|---|---|---|---|---|---|---|---|
| P | 15 | 31 | 15 | 16 | 15 | 15 | 3 | 5 |
| Ar | 18 | 40 | 18 | 22 | 18 | 18 | 3 | 8 |
Question 4: Identifying Metalloids
Metalloids have properties of both metals and nonmetals. Common metalloids: B, Si, Ge, As, Sb, Te, Po, At.
- I. Silicon (Si): Metalloid.
- II. Boron (B): Metalloid.
- III. Neon (Ne): Noble gas (nonmetal), not a metalloid.
Thus, I (Si) and II (B) are metalloids.
Question 4 Answer:
Brief Explanations
Silicon (I) and boron (II) are metalloids. Neon (III) is a noble gas (nonmetal). So only I and II are metalloids.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. I and II only