QUESTION IMAGE
Question
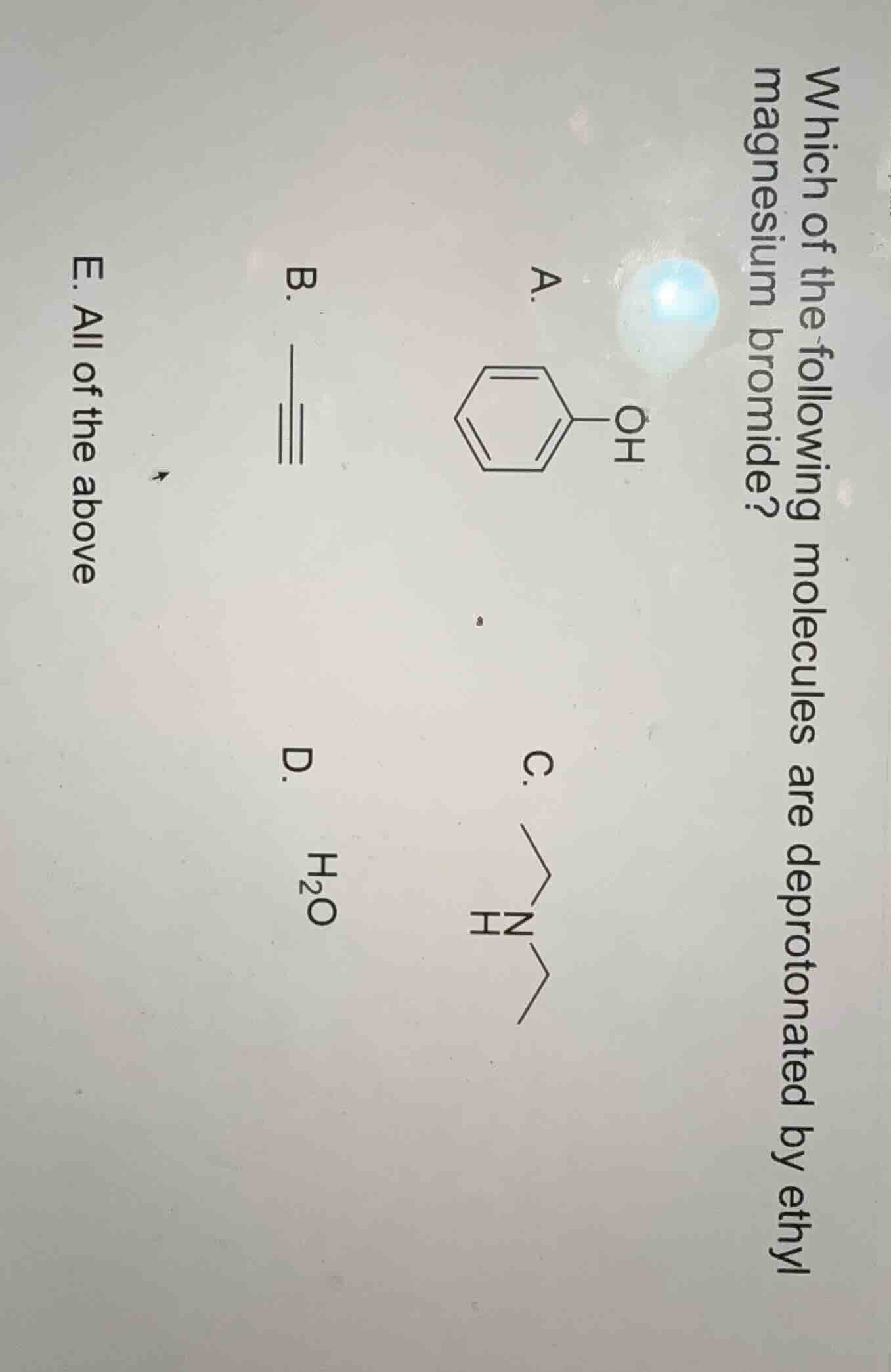
which of the following molecules are deprotonated by ethyl magnesium bromide? a. phenol structure b. alkyne structure c. diethylamine structure d. h₂o e. all of the above
Response
To determine which molecules are deprotonated by ethylmagnesium bromide (a strong base), we analyze the acidity of the protons in each molecule:
- Option A (Phenol): Phenol has a relatively acidic proton (due to resonance stabilization of the phenoxide ion). Strong bases like ethylmagnesium bromide can deprotonate phenol.
- Option B (Alkyne): Terminal alkynes have weakly acidic protons (the sp - hybridized C - H bond is relatively acidic). Ethylmagnesium bromide, as a strong base, can deprotonate terminal alkynes.
- Option C (Amine): The N - H proton in amines is weakly acidic, and strong bases like ethylmagnesium bromide can deprotonate amines.
- Option D (Water): Water has a relatively acidic proton (pKa ~ 15.7), and strong bases like ethylmagnesium bromide readily deprotonate water.
Since all the molecules (A, B, C, D) can be deprotonated by ethylmagnesium bromide, the correct answer is E.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
E. All of the above