QUESTION IMAGE
Question
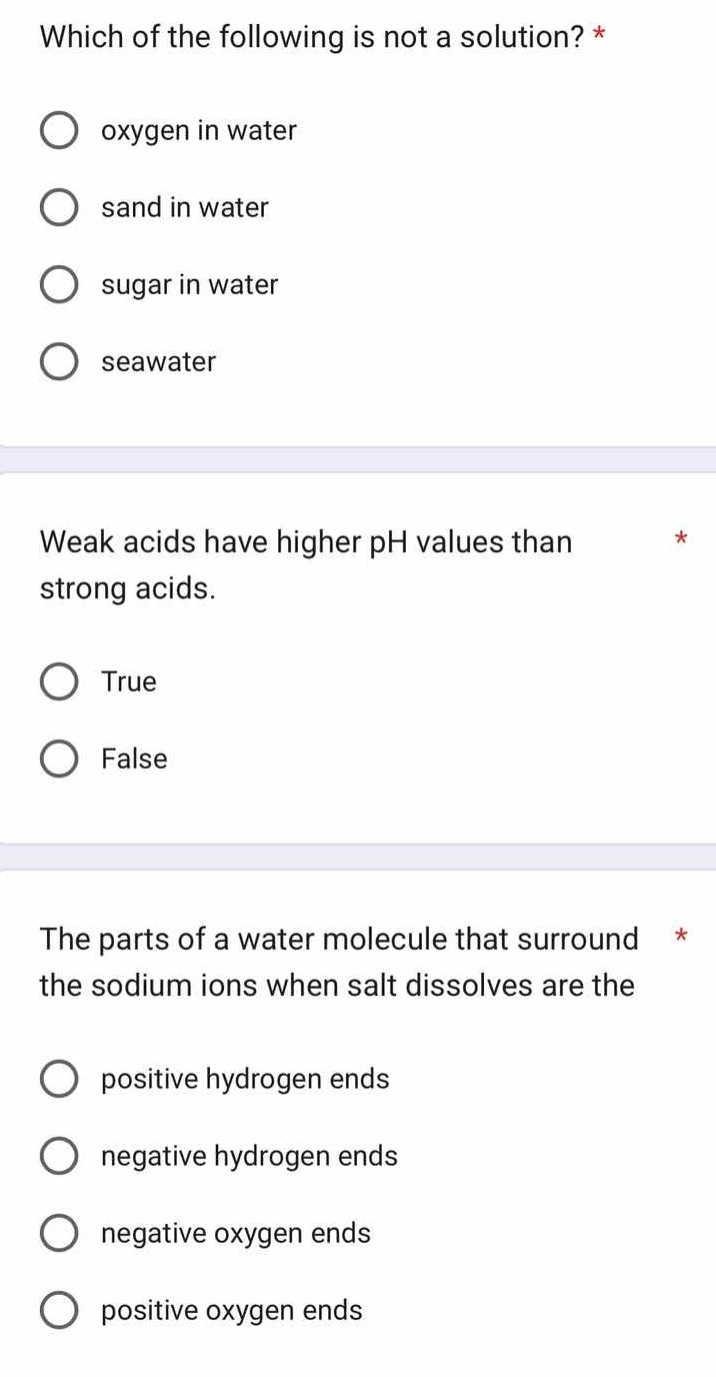
which of the following is not a solution? *
oxygen in water
sand in water
sugar in water
seawater
weak acids have higher ph values than strong acids. *
true
false
the parts of a water molecule that surround the sodium ions when salt dissolves are the *
positive hydrogen ends
negative hydrogen ends
negative oxygen ends
positive oxygen ends
Brief Explanations
- For the first question: A solution is a homogeneous mixture where the solute is evenly dispersed at the molecular level. Sand does not dissolve in water and forms a heterogeneous mixture (suspension), so it is not a solution.
- For the second question: At the same concentration, weak acids dissociate less than strong acids, producing fewer H⁺ ions, resulting in a higher pH (less acidic) compared to strong acids.
- For the third question: A water molecule is polar, with the oxygen end having a partial negative charge and hydrogen ends having partial positive charges. Sodium ions (Na⁺) are positively charged, so they are attracted to and surrounded by the negative oxygen ends of water molecules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- sand in water
- True
- negative oxygen ends