QUESTION IMAGE
Question
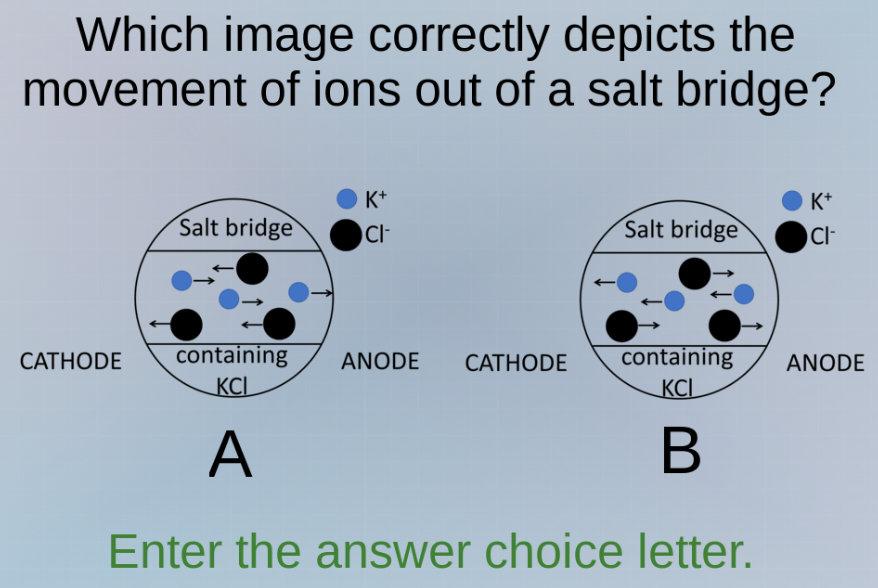
which image correctly depicts the movement of ions out of a salt bridge? salt bridge k+ ci- salt bridge k+ ci- containing containing kci kci cathode anode cathode anode a b enter the answer choice letter.
In a galvanic cell, the salt bridge (with KCl here) allows ion movement to maintain charge neutrality. At the cathode (reduction), positive ions (K⁺) move into the cathode compartment, and negative ions (Cl⁻) move out? Wait, no—wait, anode is oxidation (loses electrons, so becomes more positive), cathode is reduction (gains electrons, becomes more negative). So to balance charge: from salt bridge, K⁺ (positive) moves to cathode (to neutralize excess negative charge from reduction), and Cl⁻ (negative) moves to anode (to neutralize excess positive charge from oxidation). So in the salt bridge, K⁺ should move towards cathode (left in the diagram), Cl⁻ towards anode (right). Looking at the images: in option A, K⁺ (blue) has some moving left (towards cathode) and Cl⁻ (black) moving right (towards anode)? Wait, no—wait the salt bridge contains KCl. So inside the salt bridge, K⁺ and Cl⁻ are present. The movement: K⁺ (cation) goes to cathode (which is on the left), Cl⁻ (anion) goes to anode (right). So in the diagram, for the salt bridge's internal movement: K⁺ (blue) should move left (towards cathode side), Cl⁻ (black) move right (towards anode side). Looking at option B: K⁺ (blue) are moving left (towards cathode), Cl⁻ (black) moving right (towards anode). Wait, no—wait the "containing KCl" is the salt bridge's interior. The cathode is left, anode right. So K⁺ (positive) moves from salt bridge to cathode (left), Cl⁻ (negative) moves from salt bridge to anode (right). So inside the salt bridge, K⁺ would be moving towards cathode (left), Cl⁻ towards anode (right). So in the salt bridge's internal arrows: K⁺ (blue) arrows left, Cl⁻ (black) arrows right. Looking at the two options:
Option A: Inside salt bridge, K⁺ (blue) has some arrows left, some right? Wait no, the first image (A) has blue (K⁺) with arrows: one left, one right, one right? Wait no, let's re-examine. Wait the problem is "movement of ions out of a salt bridge"—so ions leaving the salt bridge: K⁺ out to cathode, Cl⁻ out to anode. So from the salt bridge (the circle), K⁺ (blue) should exit towards cathode (left), Cl⁻ (black) exit towards anode (right). So in the diagram, the salt bridge is the circle. So in option B: blue (K⁺) arrows left (out to cathode), black (Cl⁻) arrows right (out to anode). In option A: blue arrows: one left, one right, one right? No, maybe I got it wrong. Wait, anode is oxidation, so anode compartment becomes positive (loses electrons), so needs negative ions (Cl⁻) from salt bridge. Cathode is reduction, becomes negative, needs positive ions (K⁺) from salt bridge. So ions leaving salt bridge: K⁺ to cathode (left), Cl⁻ to anode (right). So in the salt bridge's internal movement (inside the circle), K⁺ would be moving towards cathode (left), Cl⁻ towards anode (right). So looking at the two options:
Option B: Inside the salt bridge (circle), K⁺ (blue) arrows left (towards cathode), Cl⁻ (black) arrows right (towards anode). Option A: K⁺ arrows: some left, some right, Cl⁻ arrows: some left, some right? No, maybe I messed up. Wait the correct movement is K⁺ (cation) to cathode, Cl⁻ (anion) to anode. So the ions leaving the salt bridge: K⁺ out to cathode (left), Cl⁻ out to anode (right). So in the diagram, the salt bridge is the circle. So in option B, the blue (K⁺) are moving left (out to cathode), black (Cl⁻) moving right (out to anode). So option B is correct. Wait, no—wait the first image (A) has blue (K⁺) with an arrow right (out to anode?), which is wrong. Wait, no: cathode is left, anode right. So K⁺ (positive) should go to cathode (left), Cl⁻ (negative)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B