QUESTION IMAGE
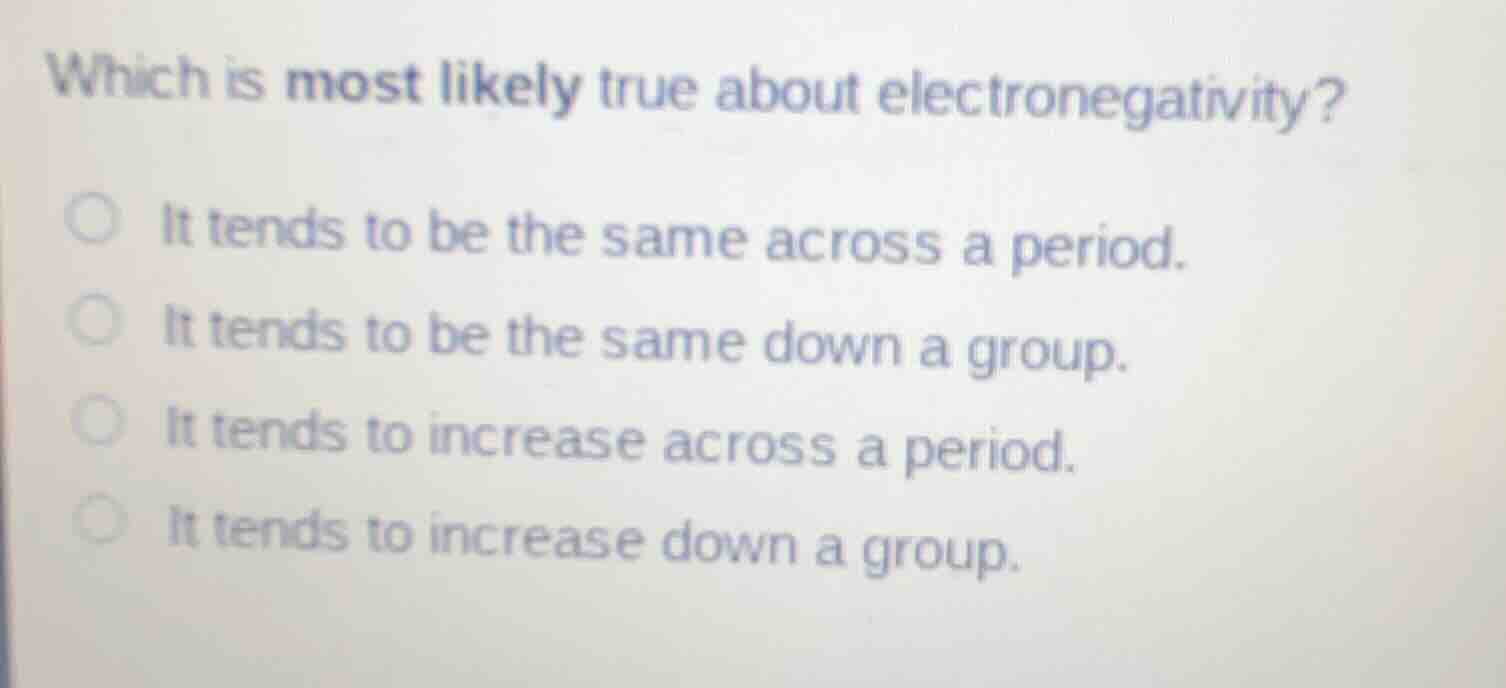
Question
which is most likely true about electronegativity?
○ it tends to be the same across a period.
○ it tends to be the same down a group.
○ it tends to increase across a period.
○ it tends to increase down a group.
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. Across a period (from left to right in the periodic table), the number of protons in the nucleus increases, and the atomic radius generally decreases. This leads to a stronger pull on electrons, so electronegativity tends to increase across a period. Down a group (from top to bottom), the atomic radius increases as new electron shells are added, and the shielding effect increases, so electronegativity tends to decrease down a group. Thus, the statement "It tends to increase across a period" is correct. The other options are incorrect: it does not stay the same across a period or down a group, and it decreases (not increases) down a group.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
It tends to increase across a period.