QUESTION IMAGE
Question
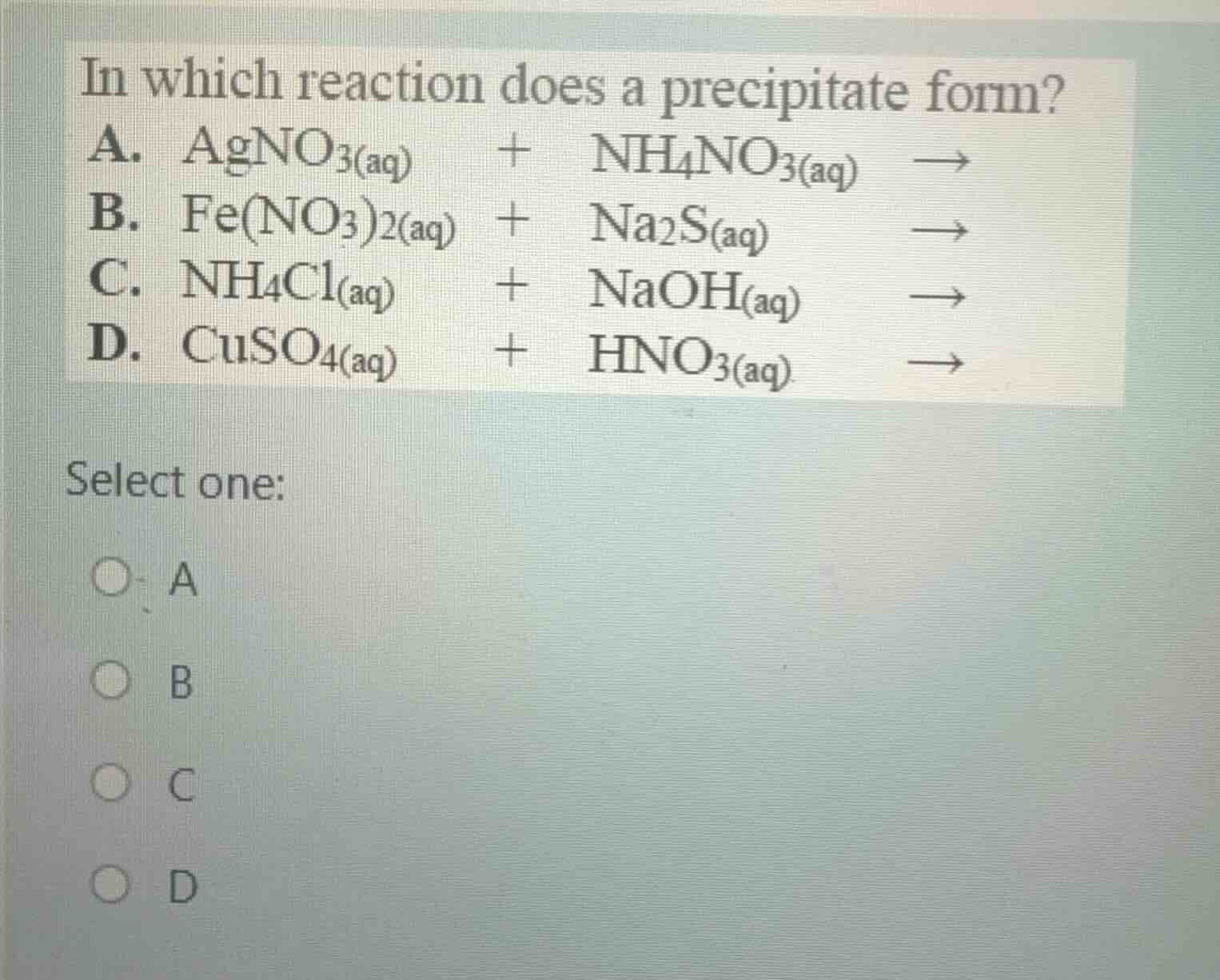
in which reaction does a precipitate form? a. agno₃(aq) + nh₄no₃(aq) → b. fe(no₃)₂(aq) + na₂s(aq) → c. nh₄cl(aq) + naoh(aq) → d. cuso₄(aq) + hno₃(aq) → select one: a b c d
Step1: Analyze Reaction A
All ions ($\text{Ag}^+$, $\text{NO}_3^-$, $\text{NH}_4^+$) remain dissolved; no precipitate forms.
Step2: Analyze Reaction B
Products are $\text{FeS}(s)$ and $\text{NaNO}_3(aq)$. $\text{FeS}$ is an insoluble sulfide precipitate.
Reaction: $\text{Fe(NO}_3\text{)}_2(aq) + \text{Na}_2\text{S}(aq)
ightarrow \text{FeS}(s) + 2\text{NaNO}_3(aq)$
Step3: Analyze Reaction C
Products are $\text{NH}_3(g)$, $\text{H}_2\text{O}(l)$, $\text{NaCl}(aq)$; no precipitate forms.
Step4: Analyze Reaction D
All ions ($\text{Cu}^{2+}$, $\text{SO}_4^{2-}$, $\text{H}^+$, $\text{NO}_3^-$) remain dissolved; no precipitate forms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{Fe(NO}_3\text{)}_2\text{(aq)} + \text{Na}_2\text{S(aq)}
ightarrow$