QUESTION IMAGE
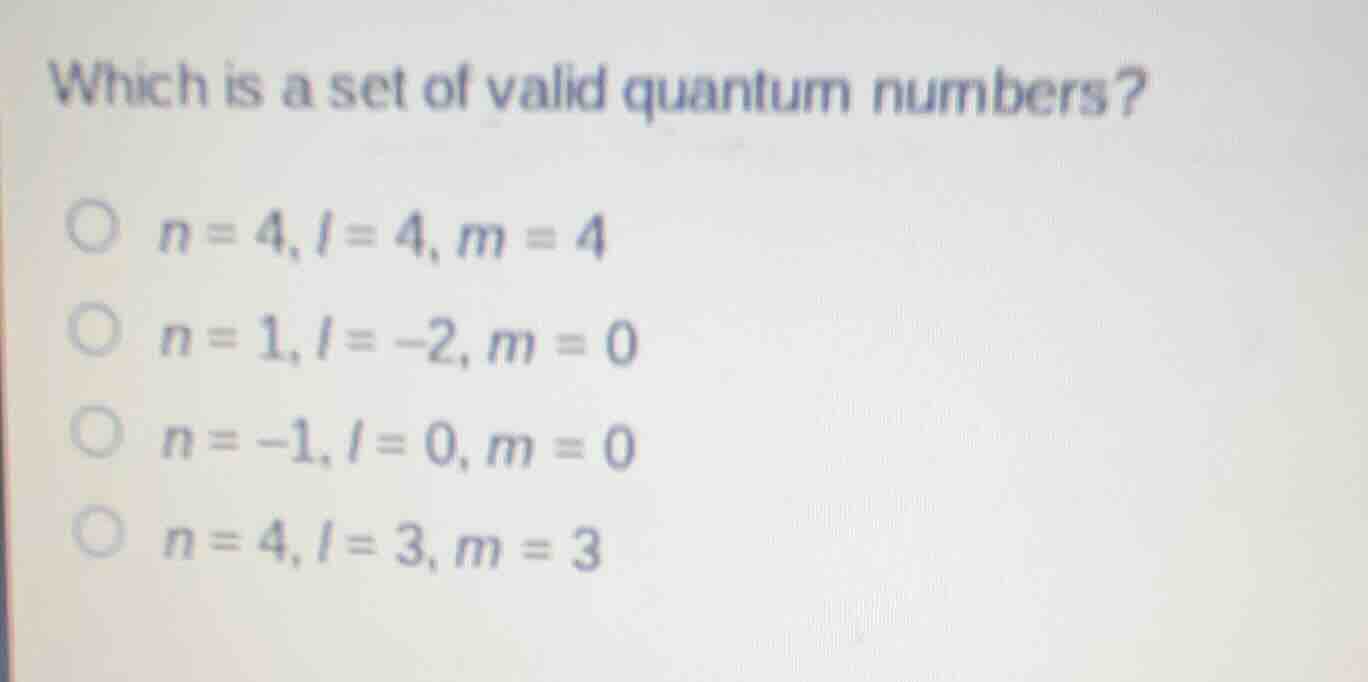
Question
which is a set of valid quantum numbers?
n = 4, l = 4, m = 4
n = 1, l = -2, m = 0
n = -1, l = 0, m = 0
n = 4, l = 3, m = 3
To determine valid quantum numbers, we use the rules for principal quantum number (\(n\)), azimuthal quantum number (\(l\)), and magnetic quantum number (\(m\)):
- \(n\) is a positive integer (\(n \geq 1\)).
- \(l\) ranges from \(0\) to \(n - 1\) (inclusive).
- \(m\) ranges from \(-l\) to \(+l\) (inclusive).
- For \(n = 4, l = 4, m = 4\):
\(l\) must be \(< n\) (since \(l\) ranges \(0\) to \(n - 1\)). Here, \(l = 4\) and \(n = 4\), so \(l = n\) is invalid.
- For \(n = 1, l = -2, m = 0\):
\(l\) cannot be negative (it ranges \(0\) to \(n - 1\)). \(n = 1\) implies \(l\) can only be \(0\), so \(l = -2\) is invalid.
- For \(n = -1, l = 0, m = 0\):
\(n\) must be a positive integer (\(n \geq 1\)). \(n = -1\) is invalid.
- For \(n = 4, l = 3, m = 3\):
- \(n = 4\) (valid, positive integer).
- \(l = 3\) (valid, since \(l\) ranges \(0\) to \(n - 1 = 3\), so \(l = 3\) is allowed).
- \(m = 3\) (valid, since \(m\) ranges from \(-l = -3\) to \(+l = 3\), so \(m = 3\) is allowed).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(n = 4, l = 3, m = 3\) (the last option)