QUESTION IMAGE
Question
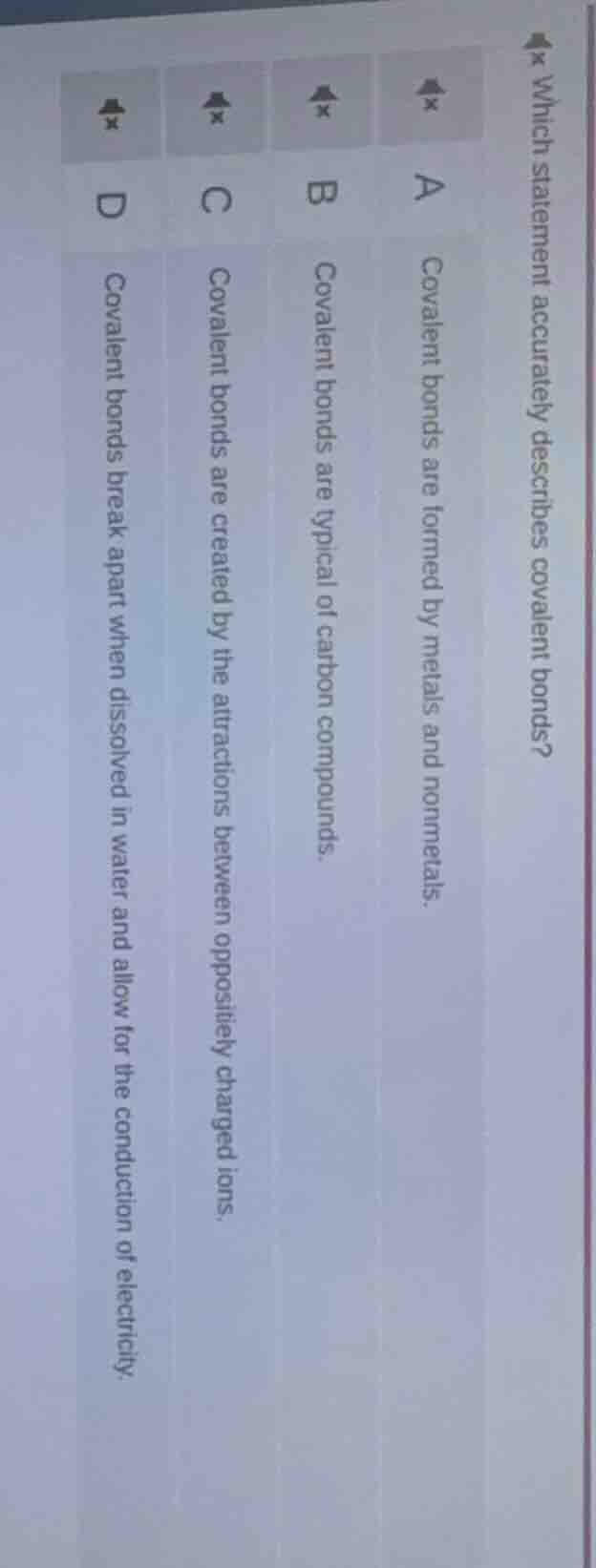
which statement accurately describes covalent bonds? a covalent bonds are formed by metals and nonmetals. b covalent bonds are typical of carbon compounds. c covalent bonds are created by the attractions between oppositely charged ions. d covalent bonds break apart when dissolved in water and allow for the conduction of electricity
Brief Explanations
- Option A: Ionic bonds are formed between metals and non - metals, not covalent bonds. So A is incorrect.
- Option B: Carbon compounds (organic compounds) are mainly held together by covalent bonds. Carbon atoms share electrons with other atoms (like H, O, N etc.) to form covalent bonds, so covalent bonds are typical of carbon compounds. This statement is correct.
- Option C: Covalent bonds are formed by the sharing of electrons between atoms, while attractions between oppositely charged ions form ionic bonds. So C is incorrect.
- Option D: Covalent compounds (like sugar, ethanol) do not break their covalent bonds when dissolved in water (they may dissociate into molecules, but covalent bonds within the molecule remain intact). Ionic compounds break ionic bonds in water for conductivity. So D is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Covalent bonds are typical of carbon compounds.