QUESTION IMAGE
Question
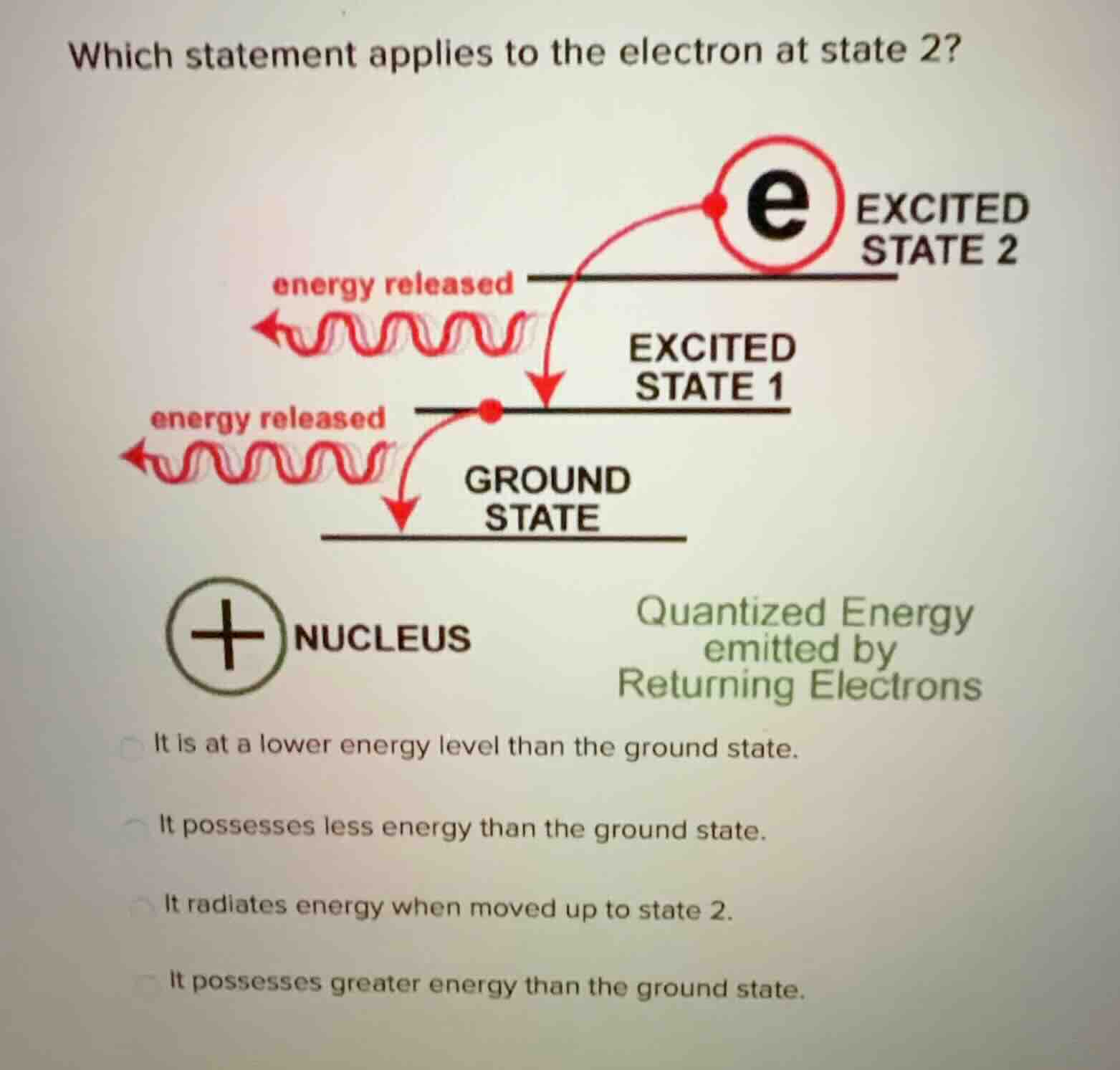
which statement applies to the electron at state 2?excited state 2excited state 1ground statenucleusquantized energy emitted by returning electronsit is at a lower energy level than the ground state.it possesses less energy than the ground state.it radiates energy when moved up to state 2.it possesses greater energy than the ground state.
Excited states have higher energy than the ground state. When electrons move to a higher excited state (like state 2), they absorb energy, not release it. The first two statements incorrectly claim state 2 has lower energy than ground state, and the third is wrong because moving to state 2 requires energy absorption, not radiation. Only the last statement matches the energy level hierarchy shown.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
It possesses greater energy than the ground state.