QUESTION IMAGE
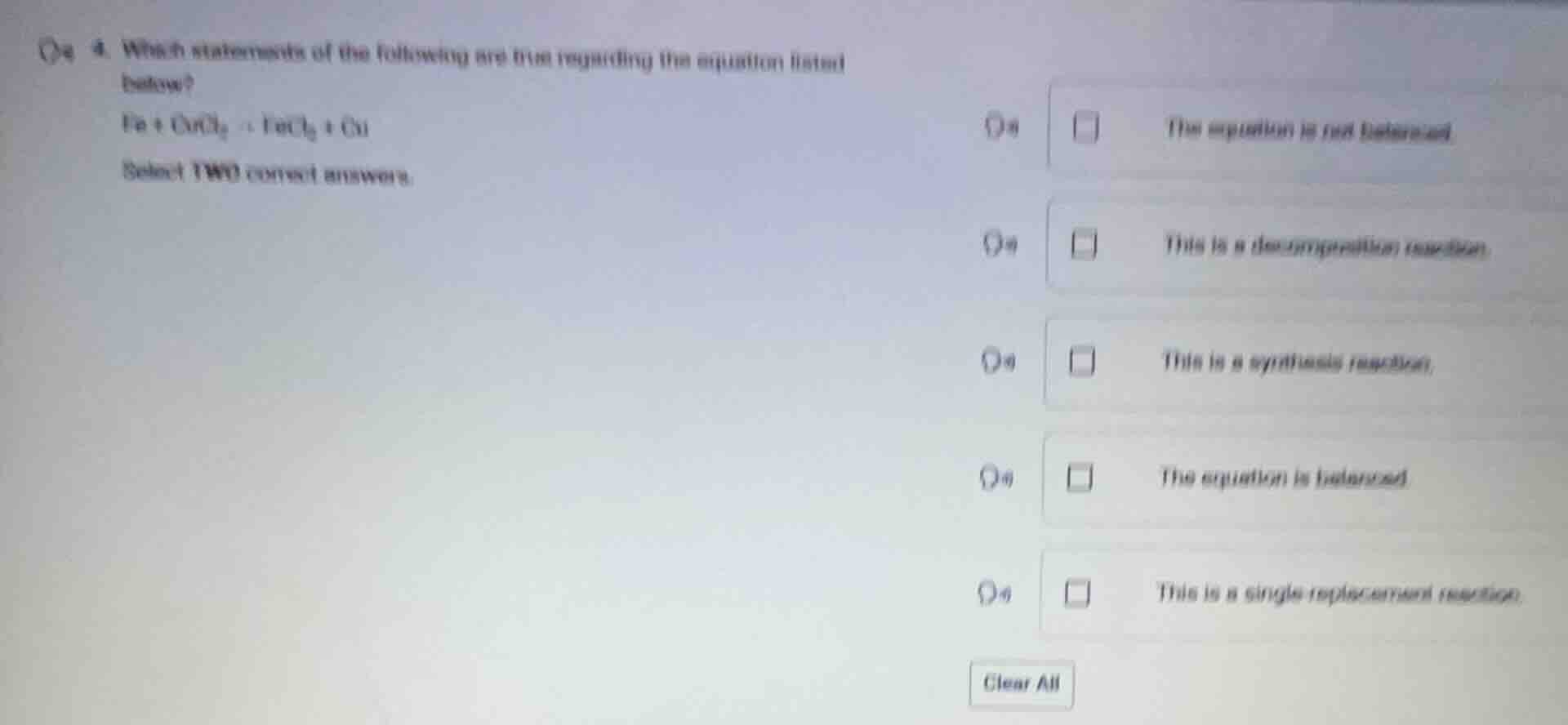
Question
- which statements of the following are true regarding the equation listed below?
fe + cucl₂ = fecl₂ + cu
select two correct answers.
☐ the equation is not balanced.
☐ this is a decomposition reaction.
☐ this is a synthesis reaction.
☐ the equation is balanced.
☐ this is a single replacement reaction.
- Balanced Check: Count atoms on both sides. Left: Fe=1, Cu=1, Cl=2. Right: Fe=1, Cu=1, Cl=2. So the equation is balanced.
- Reaction Type: In \( \text{Fe} + \text{CuCl}_2
ightarrow \text{FeCl}_2 + \text{Cu} \), Fe (a metal) replaces Cu in \( \text{CuCl}_2 \), which fits the single - replacement reaction pattern (A + BC → AC + B). Decomposition is breaking a compound into simpler substances (not the case here), and synthesis is combining substances into a compound (also not the case here).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. The equation is balanced
E. This is a single replacement reaction