QUESTION IMAGE
Question
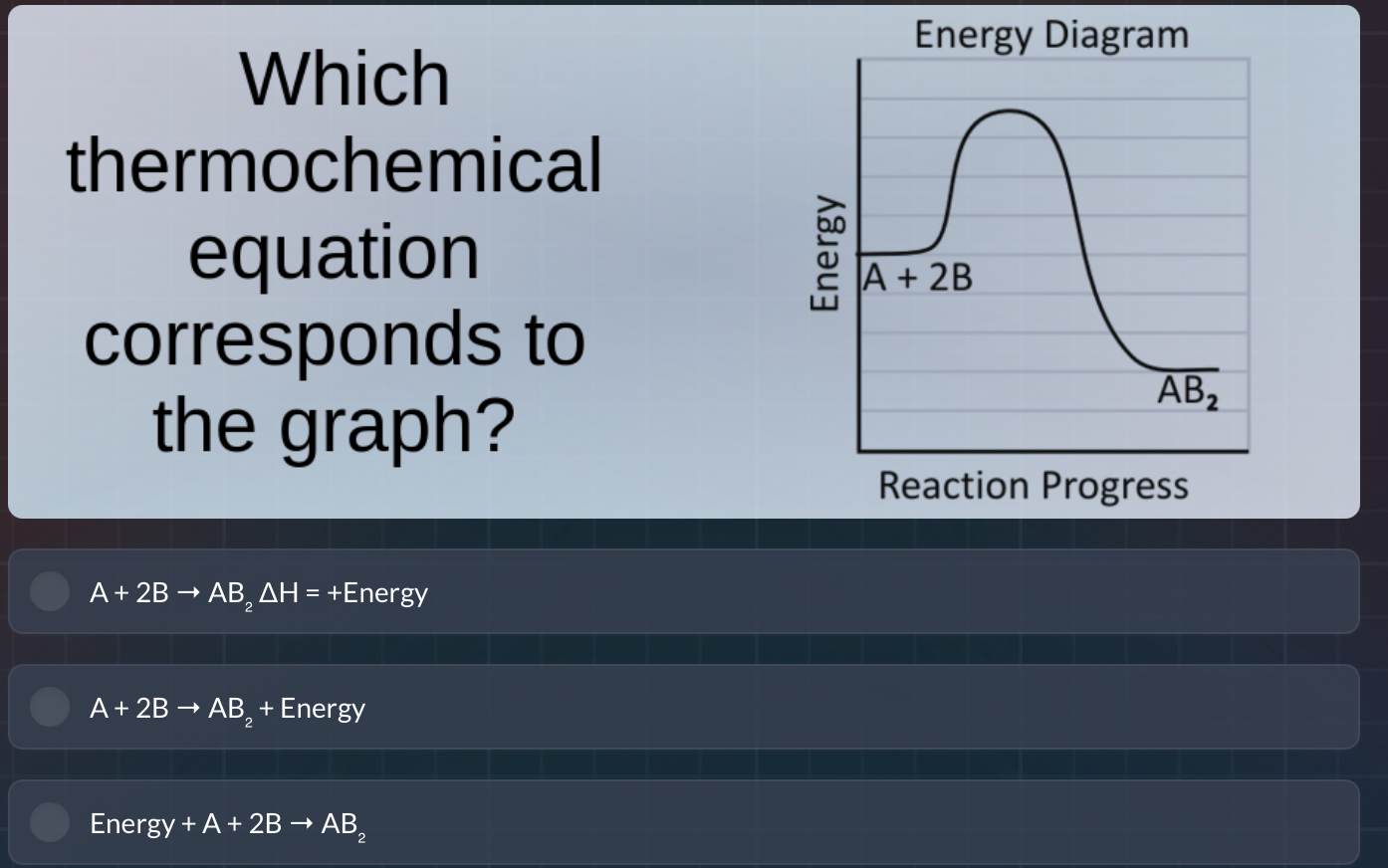
which thermochemical equation corresponds to the graph? energy diagram energy a + 2b ab₂ reaction progress a + 2b → ab₂ δh = +energy a + 2b → ab₂ + energy energy + a + 2b → ab₂
Step1: Analyze Energy Levels
The reactants (A + 2B) have higher energy than the product (AB₂) from the energy diagram.
Step2: Determine Reaction Type
In an exothermic reaction, energy is released (product energy < reactant energy). So the reaction \( \text{A} + 2\text{B}
ightarrow \text{AB}_2 \) releases energy, meaning energy is a product (added to the right side) or \( \Delta H \) is negative. The second option shows energy as a product, consistent with exothermic (energy released). The first option has \( \Delta H = +\text{Energy} \) (endothermic, wrong). The third option has energy as a reactant (endothermic, wrong).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \( \text{A} + 2\text{B}
ightarrow \text{AB}_2 + \text{Energy} \) (assuming the middle option is B, as per typical multiple - choice layout with the second option being the correct one here)