QUESTION IMAGE
Question
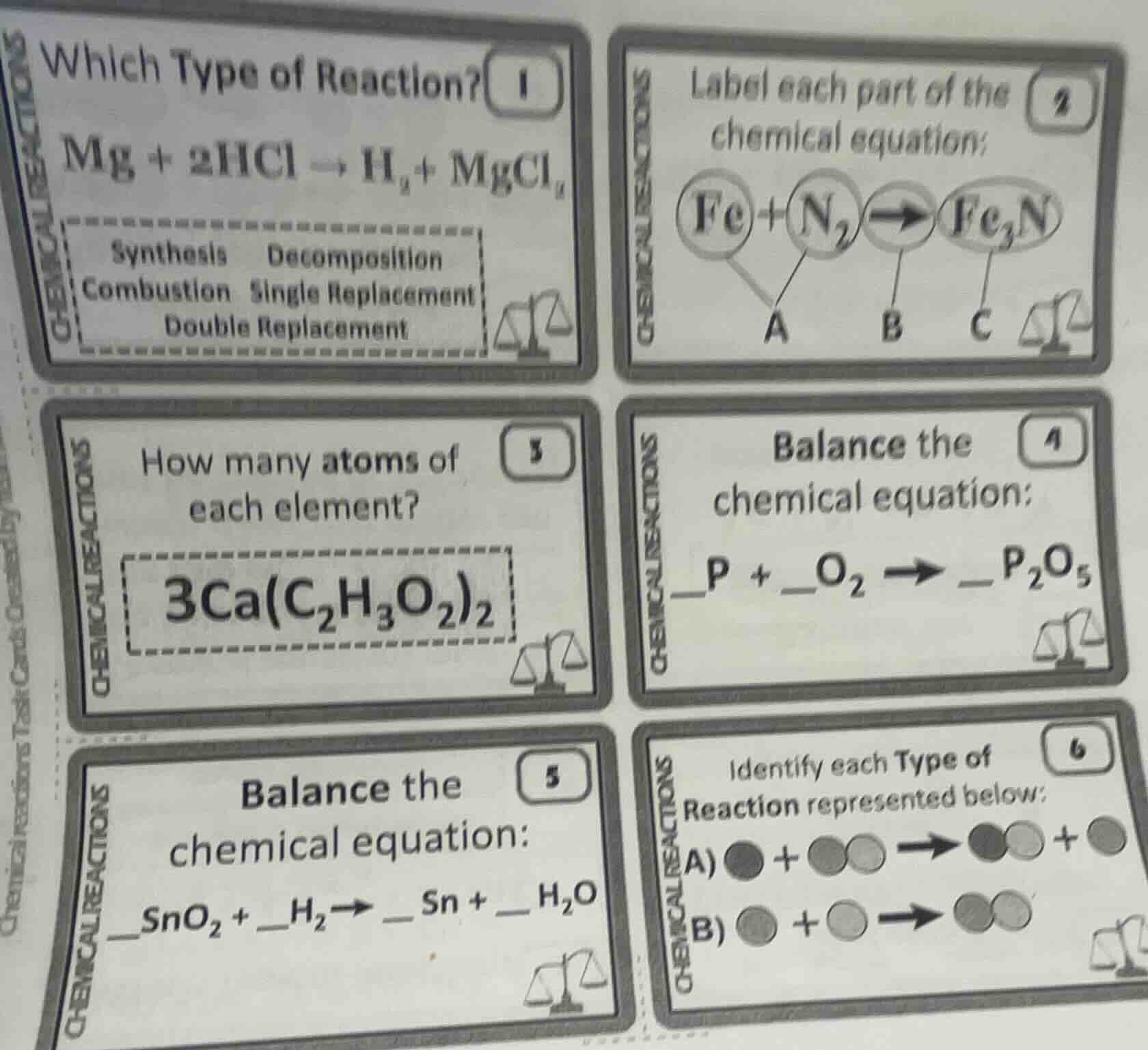
- which type of reaction? $ce{mg + 2hcl -> h_{2} + mgcl_{2}}$synthesis decompositioncombustion single replacementdouble replacement2. label each part of the chemical equation:$ce{fe + n_{2} -> fe_{3}n}$a b c3. how many atoms of each element?$ce{3ca(c_{2}h_{3}o_{2})_{2}}$4. balance the chemical equation:$ce{_p + _o_{2} -> _p_{2}o_{5}}$5. balance the chemical equation:$ce{_sno_{2} + _h_{2} -> _sn + _h_{2}o}$6. identify each type of reaction represented below:a) ● + ●○ → ●○ + ●b) ● + ○ → ●○
Step1: Identify reaction type (Q1)
In $\text{Mg} + 2\text{HCl}
ightarrow \text{H}_2 + \text{MgCl}_2$, a single element (Mg) replaces another element (H) in a compound (HCl). This matches single replacement.
Step2: Label equation parts (Q2)
A: Reactants (starting substances: $\text{Fe}, \text{N}_2$); B: Yield sign (shows reaction direction); C: Product (substance formed: $\text{Fe}_3\text{N}$)
Step3: Count atoms (Q3)
For $3\text{Ca(C}_2\text{H}_3\text{O}_2\text{)}_2$:
- Ca: $3 \times 1 = 3$
- C: $3 \times (2 \times 2) = 12$
- H: $3 \times (3 \times 2) = 18$
- O: $3 \times (2 \times 2) = 12$
Step4: Balance equation (Q4)
Balance $\text{P} + \text{O}_2
ightarrow \text{P}_2\text{O}_5$:
- Balance P: $2\text{P} + \text{O}_2
ightarrow \text{P}_2\text{O}_5$
- Balance O: $2\text{P} + \frac{5}{2}\text{O}_2
ightarrow \text{P}_2\text{O}_5$, multiply all by 2: $4\text{P} + 5\text{O}_2
ightarrow 2\text{P}_2\text{O}_5$
Step5: Balance equation (Q5)
Balance $\text{SnO}_2 + \text{H}_2
ightarrow \text{Sn} + \text{H}_2\text{O}$:
- Balance O: $\text{SnO}_2 + \text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
- Balance H: $\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
Step6: Identify reaction types (Q6)
A: One element replaces another in a compound → Single Replacement; B: Two substances form one compound → Synthesis
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Single Replacement
- A: Reactants; B: Yield Sign; C: Product
- Ca: 3 atoms, C: 12 atoms, H: 18 atoms, O: 12 atoms
- $4\text{P} + 5\text{O}_2
ightarrow 2\text{P}_2\text{O}_5$ (coefficients: 4, 5, 2)
- $\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$ (coefficients: 1, 2, 1, 2)
- A: Single Replacement; B: Synthesis