QUESTION IMAGE
Question
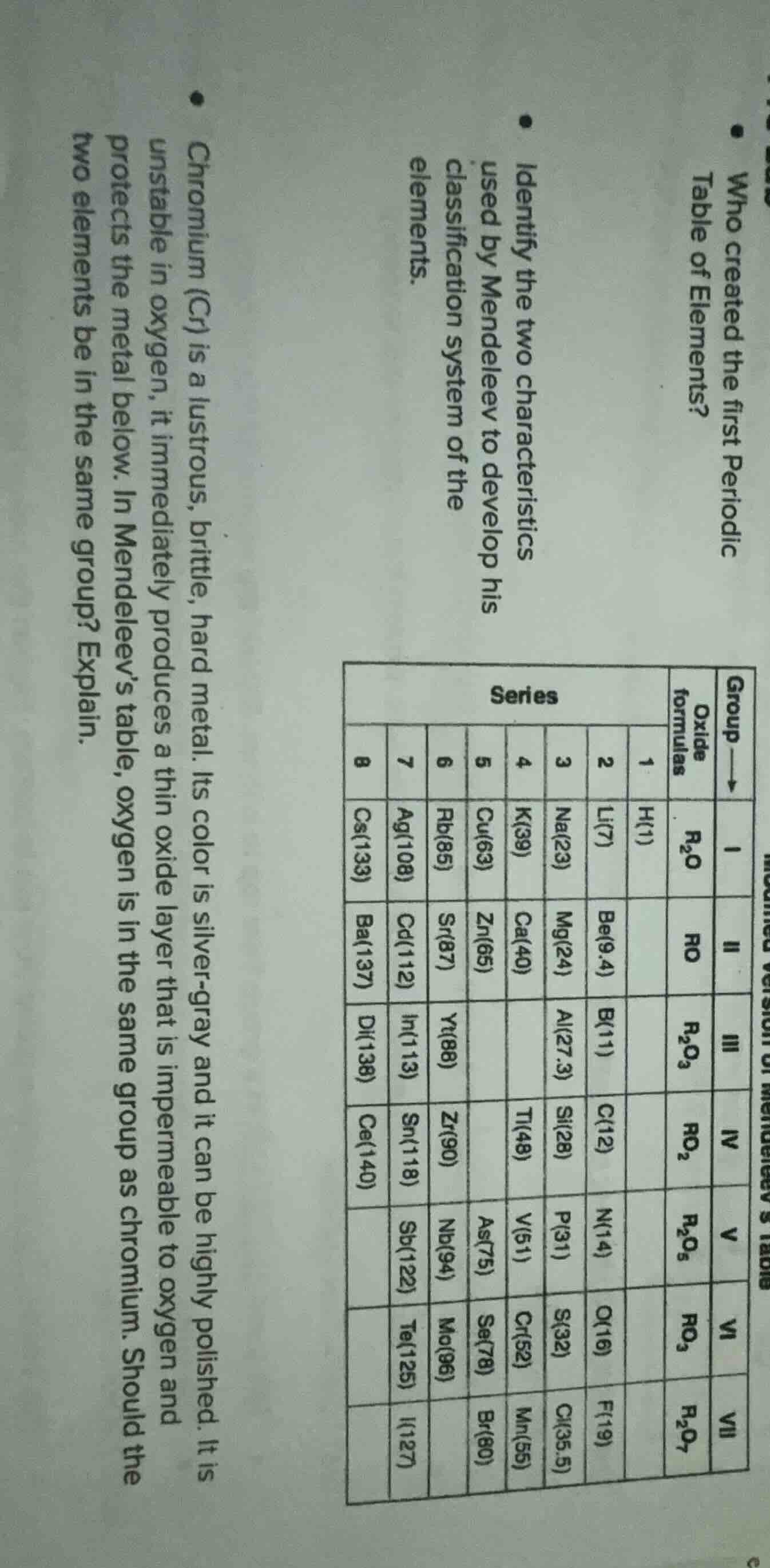
who created the first periodic table of elements? identify the two characteristics used by mendeleev to develop his classification system of the elements. chromium (cr) is a lustrous, brittle, hard metal. its color is silver - gray and it can be highly polished. it is unstable in oxygen, it immediately produces a thin oxide layer that is impermeable to oxygen and protects the metal below. in mendeleev’s table, oxygen is in the same group as chromium. should the two elements be in the same group? explain. and there is a table about group, oxide formulas and series with elements like h, li, na, etc. and their oxide formulas and series numbers
Part 1: Identify the two characteristics used by Mendeleev
Mendeleev used atomic mass (the numbers in parentheses, e.g., H(1), Li(7)) and chemical properties (reflected by oxide formulas, e.g., \( R_2O \), \( RO \)) to develop his periodic table. Atomic mass ordered elements, and chemical properties (like oxide formation) grouped elements with similar reactivity.
In Mendeleev’s table, elements in the same group have the same oxide formula pattern (chemical property) and similar valence. Chromium (Cr) has an oxide formula pattern matching Group VI (\( RO_3 \), as Cr forms \( CrO_3 \) or similar oxides with 6+ valence). Oxygen (O) is in Group VI (oxide formula \( RO_3 \) for the group, and O’s valence in oxides aligns with Group VI). Since they share the same group’s oxide formula pattern (chemical property) and valence, they should be in the same group.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Atomic Mass (from the numbers in parentheses for each element).
- Chemical Properties (from oxide formulas, e.g., \( R_2O \), \( RO \), \( R_2O_3 \), etc., showing similar reactivity).