QUESTION IMAGE
Question
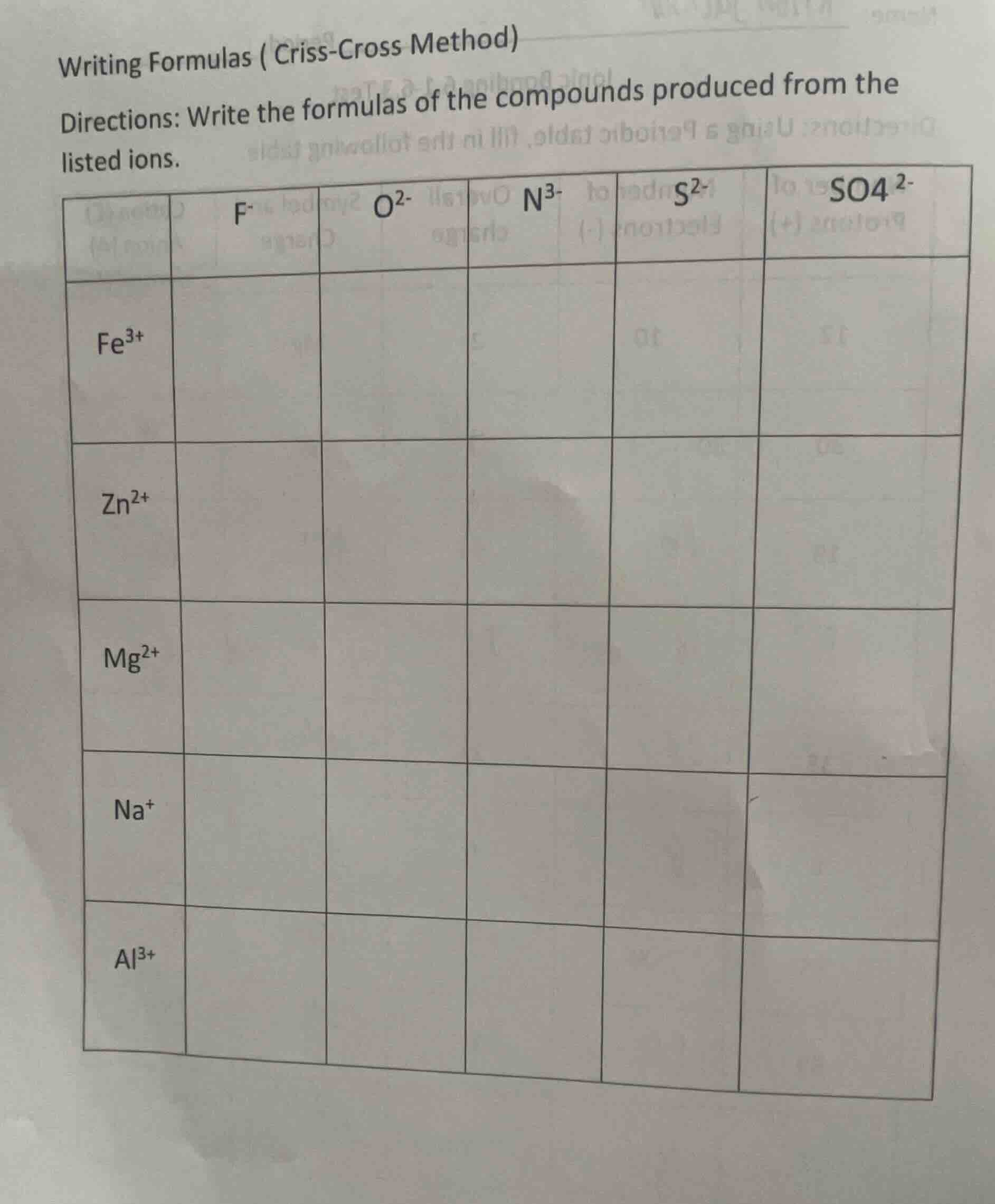
writing formulas ( criss-cross method)directions: write the formulas of the compounds produced from the listed ions.$f^-$ $o^{2-}$ $n^{3-}$ $s^{2-}$ $so4^{2-}$$fe^{3+}$$zn^{2+}$$mg^{2+}$$na^+$$al^{3+}$
Step1: Criss-cross charges for $\text{Fe}^{3+}$
- With $\text{F}^-$: $\text{Fe}^{3+}$ and $\text{F}^-$ → $\text{FeF}_3$
- With $\text{O}^{2-}$: $\text{Fe}^{3+}$ and $\text{O}^{2-}$ → $\text{Fe}_2\text{O}_3$
- With $\text{N}^{3-}$: $\text{Fe}^{3+}$ and $\text{N}^{3-}$ → $\text{FeN}$
- With $\text{S}^{2-}$: $\text{Fe}^{3+}$ and $\text{S}^{2-}$ → $\text{Fe}_2\text{S}_3$
- With $\text{SO}_4^{2-}$: $\text{Fe}^{3+}$ and $\text{SO}_4^{2-}$ → $\text{Fe}_2(\text{SO}_4)_3$
Step2: Criss-cross charges for $\text{Zn}^{2+}$
- With $\text{F}^-$: $\text{Zn}^{2+}$ and $\text{F}^-$ → $\text{ZnF}_2$
- With $\text{O}^{2-}$: $\text{Zn}^{2+}$ and $\text{O}^{2-}$ → $\text{ZnO}$
- With $\text{N}^{3-}$: $\text{Zn}^{2+}$ and $\text{N}^{3-}$ → $\text{Zn}_3\text{N}_2$
- With $\text{S}^{2-}$: $\text{Zn}^{2+}$ and $\text{S}^{2-}$ → $\text{ZnS}$
- With $\text{SO}_4^{2-}$: $\text{Zn}^{2+}$ and $\text{SO}_4^{2-}$ → $\text{ZnSO}_4$
Step3: Criss-cross charges for $\text{Mg}^{2+}$
- With $\text{F}^-$: $\text{Mg}^{2+}$ and $\text{F}^-$ → $\text{MgF}_2$
- With $\text{O}^{2-}$: $\text{Mg}^{2+}$ and $\text{O}^{2-}$ → $\text{MgO}$
- With $\text{N}^{3-}$: $\text{Mg}^{2+}$ and $\text{N}^{3-}$ → $\text{Mg}_3\text{N}_2$
- With $\text{S}^{2-}$: $\text{Mg}^{2+}$ and $\text{S}^{2-}$ → $\text{MgS}$
- With $\text{SO}_4^{2-}$: $\text{Mg}^{2+}$ and $\text{SO}_4^{2-}$ → $\text{MgSO}_4$
Step4: Criss-cross charges for $\text{Na}^+$
- With $\text{F}^-$: $\text{Na}^+$ and $\text{F}^-$ → $\text{NaF}$
- With $\text{O}^{2-}$: $\text{Na}^+$ and $\text{O}^{2-}$ → $\text{Na}_2\text{O}$
- With $\text{N}^{3-}$: $\text{Na}^+$ and $\text{N}^{3-}$ → $\text{Na}_3\text{N}$
- With $\text{S}^{2-}$: $\text{Na}^+$ and $\text{S}^{2-}$ → $\text{Na}_2\text{S}$
- With $\text{SO}_4^{2-}$: $\text{Na}^+$ and $\text{SO}_4^{2-}$ → $\text{Na}_2\text{SO}_4$
Step5: Criss-cross charges for $\text{Al}^{3+}$
- With $\text{F}^-$: $\text{Al}^{3+}$ and $\text{F}^-$ → $\text{AlF}_3$
- With $\text{O}^{2-}$: $\text{Al}^{3+}$ and $\text{O}^{2-}$ → $\text{Al}_2\text{O}_3$
- With $\text{N}^{3-}$: $\text{Al}^{3+}$ and $\text{N}^{3-}$ → $\text{AlN}$
- With $\text{S}^{2-}$: $\text{Al}^{3+}$ and $\text{S}^{2-}$ → $\text{Al}_2\text{S}_3$
- With $\text{SO}_4^{2-}$: $\text{Al}^{3+}$ and $\text{SO}_4^{2-}$ → $\text{Al}_2(\text{SO}_4)_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| $\text{F}^-$ | $\text{O}^{2-}$ | $\text{N}^{3-}$ | $\text{S}^{2-}$ | $\text{SO}_4^{2-}$ | |
|---|---|---|---|---|---|
| $\text{Zn}^{2+}$ | $\text{ZnF}_2$ | $\text{ZnO}$ | $\text{Zn}_3\text{N}_2$ | $\text{ZnS}$ | $\text{ZnSO}_4$ |
| $\text{Mg}^{2+}$ | $\text{MgF}_2$ | $\text{MgO}$ | $\text{Mg}_3\text{N}_2$ | $\text{MgS}$ | $\text{MgSO}_4$ |
| $\text{Na}^+$ | $\text{NaF}$ | $\text{Na}_2\text{O}$ | $\text{Na}_3\text{N}$ | $\text{Na}_2\text{S}$ | $\text{Na}_2\text{SO}_4$ |
| $\text{Al}^{3+}$ | $\text{AlF}_3$ | $\text{Al}_2\text{O}_3$ | $\text{AlN}$ | $\text{Al}_2\text{S}_3$ | $\text{Al}_2(\text{SO}_4)_3$ |