QUESTION IMAGE
Question
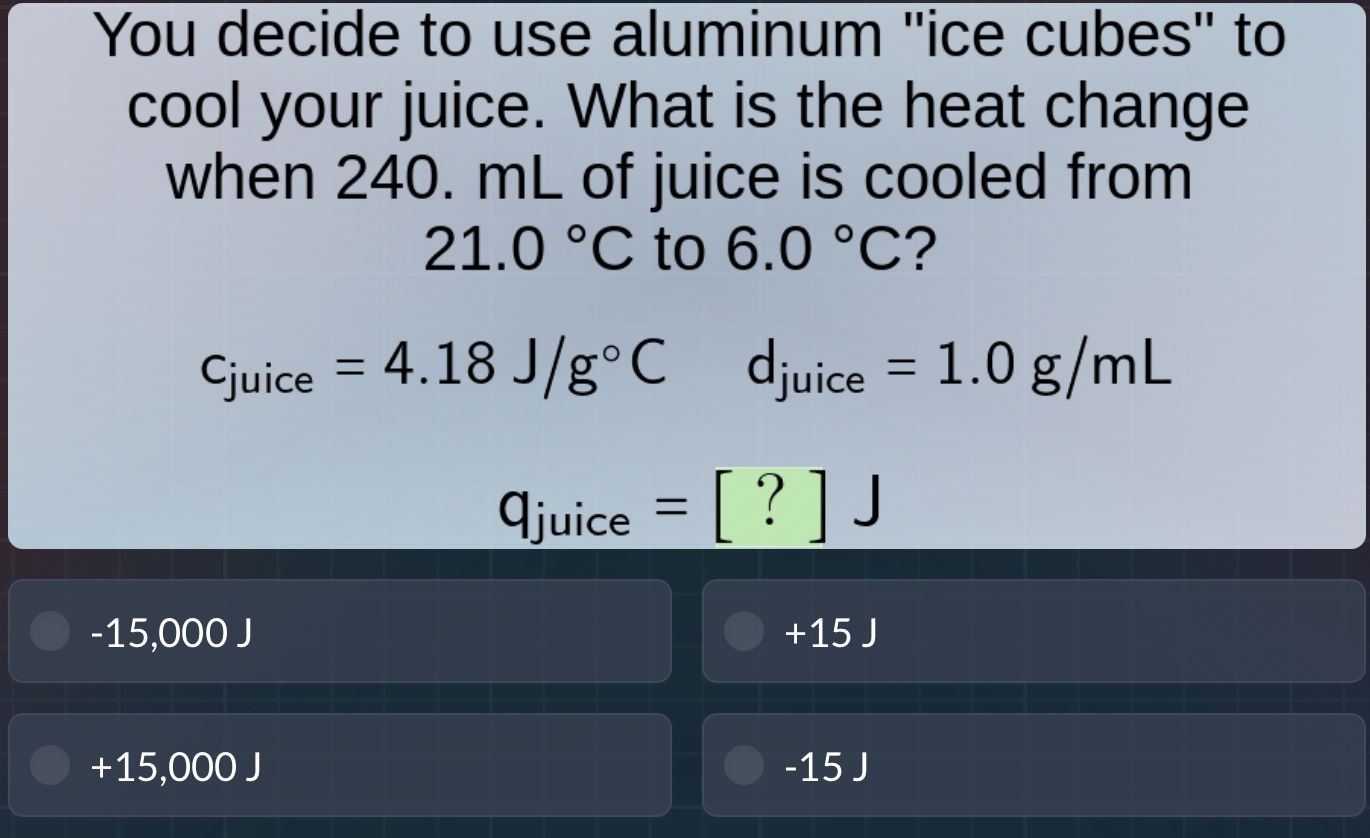
you decide to use aluminum \ice cubes\ to cool your juice. what is the heat change when 240. ml of juice is cooled from 21.0 °c to 6.0 °c?
$c_{juice} = 4.18 \text{ j/g°}c$ $d_{juice} = 1.0 \text{ g/ml}$
$q_{juice} = ? \text{ j}$
-15,000 j +15 j
+15,000 j -15 j
Step1: Calculate mass of juice
Using density formula \(
ho = \frac{m}{V} \), rearranged to \( m =
ho V \).
Given \(
ho = 1.0 \, \text{g/mL} \), \( V = 240. \, \text{mL} \), so \( m = 1.0 \, \text{g/mL} \times 240. \, \text{mL} = 240. \, \text{g} \).
Step2: Calculate temperature change
\( \Delta T = T_{\text{final}} - T_{\text{initial}} = 6.0^\circ\text{C} - 21.0^\circ\text{C} = -15.0^\circ\text{C} \).
Step3: Calculate heat change (\( q \))
Using \( q = mc\Delta T \), where \( m = 240. \, \text{g} \), \( c = 4.18 \, \text{J/g}^\circ\text{C} \), \( \Delta T = -15.0^\circ\text{C} \).
\( q = 240. \, \text{g} \times 4.18 \, \text{J/g}^\circ\text{C} \times (-15.0^\circ\text{C}) \).
First, \( 240 \times 4.18 = 1003.2 \), then \( 1003.2 \times (-15) = -15048 \, \text{J} \approx -15,000 \, \text{J} \) (rounded).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
-15,000 J