QUESTION IMAGE
Question
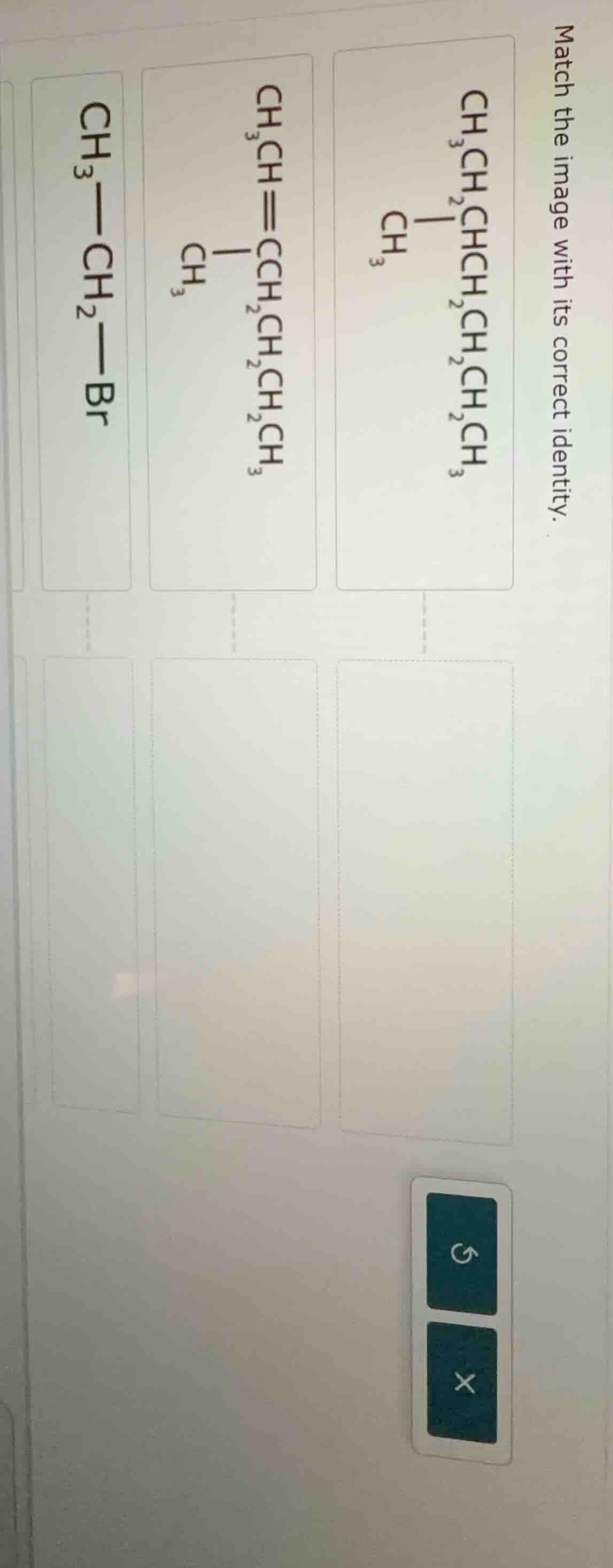
match the image with its correct identity.
Response
To solve this, we analyze each structure:
First Structure (\(\boldsymbol{\ce{CH3CH2CHCH2CH2CH2CHCH3}}\) with a \(\ce{CH3}\) branch)
- Count carbon atoms: The main chain (longest continuous C chain) has 8 carbons (parent chain: octane). The branch is a \(\ce{CH3}\) at carbon 3 (numbering from left, the branch is on the 3rd C). So it’s 3 - methyloctane (alkane, single bonds only).
Second Structure (\(\boldsymbol{\ce{CH3CH=CHCH2CH2CH2CHCH3}}\) with a \(\ce{CH3}\) branch)
- The double bond (\(\ce{C=C}\)) is at carbon 2 - 3 (numbering to give double bond lowest number). The main chain has 8 carbons (parent chain: octene, since it has a double bond). The branch is a \(\ce{CH3}\) at carbon 7. So it’s 7 - methyl - 2 - octene (alkene, has a \(\ce{C=C}\) bond).
Third Structure (\(\boldsymbol{\ce{CH3 - CH2 - Br}}\))
- This is a two - carbon chain (\(\ce{C2H5}\)) with a \(\ce{Br}\) substituent. So it’s bromoethane (alkyl halide, has a halogen (\(\ce{Br}\)) attached to an alkane chain).
If we assume the “identities” to match are likely alkane, alkene, and alkyl halide, we pair:
- The first (alkane, single bonds) → 3 - methyloctane (or “octane with methyl branch”).
- The second (alkene, double bond) → 7 - methyl - 2 - octene (or “octene with methyl and double bond”).
- The third (alkyl halide) → bromoethane (or “ethyl bromide”).
(Note: If the “identities” are labels like “alkane”, “alkene”, “alkyl halide”, the matches are:
- First structure: Alkane (3 - methyloctane)
- Second structure: Alkene (7 - methyl - 2 - octene)
- Third structure: Alkyl halide (bromoethane))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each structure:
First Structure (\(\boldsymbol{\ce{CH3CH2CHCH2CH2CH2CHCH3}}\) with a \(\ce{CH3}\) branch)
- Count carbon atoms: The main chain (longest continuous C chain) has 8 carbons (parent chain: octane). The branch is a \(\ce{CH3}\) at carbon 3 (numbering from left, the branch is on the 3rd C). So it’s 3 - methyloctane (alkane, single bonds only).
Second Structure (\(\boldsymbol{\ce{CH3CH=CHCH2CH2CH2CHCH3}}\) with a \(\ce{CH3}\) branch)
- The double bond (\(\ce{C=C}\)) is at carbon 2 - 3 (numbering to give double bond lowest number). The main chain has 8 carbons (parent chain: octene, since it has a double bond). The branch is a \(\ce{CH3}\) at carbon 7. So it’s 7 - methyl - 2 - octene (alkene, has a \(\ce{C=C}\) bond).
Third Structure (\(\boldsymbol{\ce{CH3 - CH2 - Br}}\))
- This is a two - carbon chain (\(\ce{C2H5}\)) with a \(\ce{Br}\) substituent. So it’s bromoethane (alkyl halide, has a halogen (\(\ce{Br}\)) attached to an alkane chain).
If we assume the “identities” to match are likely alkane, alkene, and alkyl halide, we pair:
- The first (alkane, single bonds) → 3 - methyloctane (or “octane with methyl branch”).
- The second (alkene, double bond) → 7 - methyl - 2 - octene (or “octene with methyl and double bond”).
- The third (alkyl halide) → bromoethane (or “ethyl bromide”).
(Note: If the “identities” are labels like “alkane”, “alkene”, “alkyl halide”, the matches are:
- First structure: Alkane (3 - methyloctane)
- Second structure: Alkene (7 - methyl - 2 - octene)
- Third structure: Alkyl halide (bromoethane))