QUESTION IMAGE
Question
name:
mad minute: vsepr shapes & angles
directions: fill in the missing information with diagrams, shapes, angles or bonding pair and lone pairs.
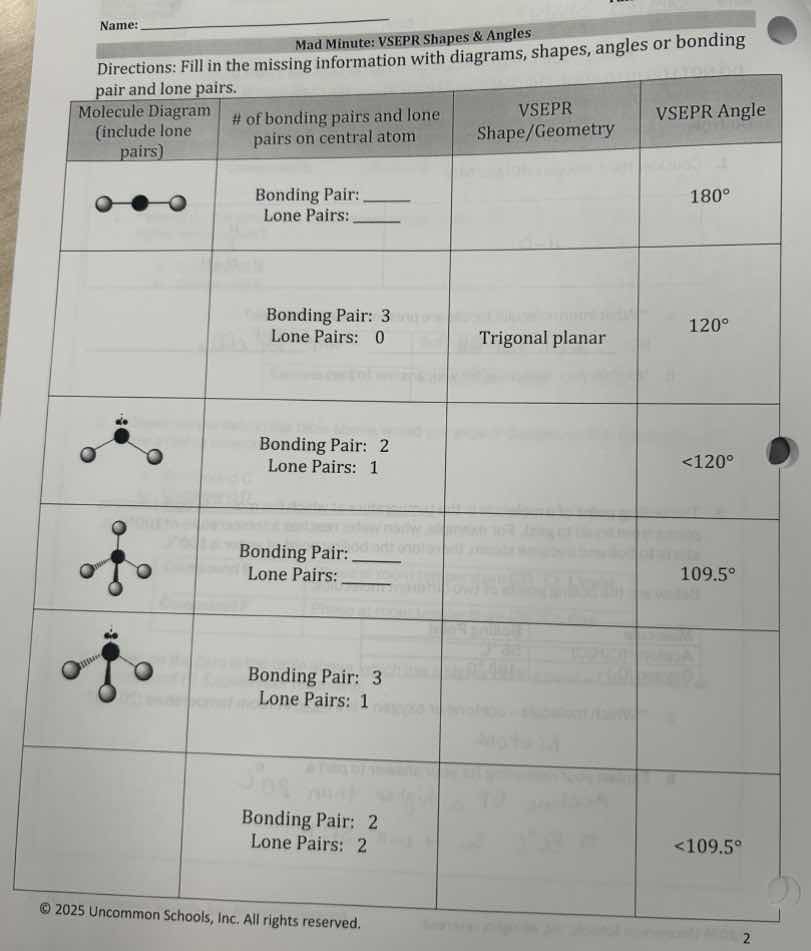
| molecule diagram (include lone pairs) | # of bonding pairs and lone pairs on central atom | vsepr shape/geometry | vsepr angle |
|---|
| diagram of a linear molecule | bonding pair: ____
lone pairs: ____ | | 180° |
| | bonding pair: 3
lone pairs: 0 | trigonal planar | 120° |
| diagram of a molecule with 2 bonding pairs and 1 lone pair | bonding pair: 2
lone pairs: 1 | | <120° |
| diagram of a tetrahedral molecule | bonding pair: ____
lone pairs: ____ | | 109.5° |
| diagram of a molecule with 3 bonding pairs and 1 lone pair | bonding pair: 3
lone pairs: 1 | | |
| | bonding pair: 2
lone pairs: 2 | | <109.5° |
© 2025 uncommon schools, inc. all rights reserved.
2
First Row (Linear Molecule):
Step1: Analyze the molecule diagram
The first molecule diagram is a straight line with three atoms (two terminal, one central), so it's a linear structure. For a linear molecule, the central atom has 2 bonding pairs and 0 lone pairs (since there are no lone pairs shown, and the geometry is linear which corresponds to 2 bonding pairs and 0 lone pairs in VSEPR).
Bonding Pair: 2
Lone Pairs: 0
VSEPR Shape/Geometry: Linear
Second Row (Trigonal Planar):
The molecule diagram for trigonal planar (with 3 bonding pairs and 0 lone pairs) would be a central atom with three terminal atoms forming a triangle (planar) around it. So we can draw a central atom with three atoms attached, each at 120° to each other.
Third Row (Bent or V - shaped with 1 lone pair):
The molecule diagram has a central atom with two terminal atoms and one lone pair (represented by the two dots). The VSEPR shape here is bent (or angular) because of the 1 lone pair. So VSEPR Shape/Geometry: Bent (or Angular)
Fourth Row (Tetrahedral - like with 4 bonding pairs):
The molecule diagram has a central atom with four terminal atoms, which is a tetrahedral geometry. In tetrahedral, the central atom has 4 bonding pairs and 0 lone pairs (since all electron pairs are bonding, as there are no lone pairs shown).
Bonding Pair: 4
Lone Pairs: 0
VSEPR Shape/Geometry: Tetrahedral
Fifth Row (Trigonal Pyramidal):
The molecule diagram has a central atom with three terminal atoms and one lone pair (represented by the two dots). The VSEPR shape here is trigonal pyramidal (because of the 1 lone pair, the shape is a pyramid with a triangular base).
VSEPR Shape/Geometry: Trigonal Pyramidal
The VSEPR angle here is less than 109.5° (since lone pairs repel more than bonding pairs, compressing the angle between the bonding pairs).
Sixth Row (Bent or V - shaped with 2 lone pairs):
The molecule has 2 bonding pairs and 2 lone pairs. The VSEPR shape here is bent (or angular) with a tetrahedral electron - pair geometry but molecular geometry is bent. The molecule diagram would have a central atom with two terminal atoms and two lone pairs (represented by dots).
VSEPR Shape/Geometry: Bent (or Angular)
Now, filling in the table:
| Molecule Diagram (include lone pairs) | # of bonding pairs and lone pairs on central atom | VSEPR Shape/Geometry | VSEPR Angle |
|---|---|---|---|
| <img src="https://via.placeholder.com/80x40?text=Trigonal+Planar" /> (trigonal planar, 3 bonding, 0 lone) | Bonding Pair: 3<br>Lone Pairs: 0 | Trigonal planar | $120^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+1+Lone+Pair" /> (bent, 2 bonding, 1 lone) | Bonding Pair: 2<br>Lone Pairs: 1 | Bent (Angular) | $<120^{\circ}$ |
| <img src="https://via.placeholder.com/60x40?text=Tetrahedral" /> (tetrahedral, 4 bonding, 0 lone) | Bonding Pair: 4<br>Lone Pairs: 0 | Tetrahedral | $109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Trigonal+Pyramidal" /> (trigonal pyramidal, 3 bonding, 1 lone) | Bonding Pair: 3<br>Lone Pairs: 1 | Trigonal Pyramidal | $<109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+2+Lone+Pairs" /> (bent, 2 bonding, 2 lone) | Bonding Pair: 2<br>Lone Pairs: 2 | Bent (Angular) | $<109.5^{\circ}$ |
Final Table:
| Molecule Diagram (include lone pairs) | # of bonding pairs and lone pairs on central atom | VSEPR Shape/Geometry | VSEPR Angle… |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First Row (Linear Molecule):
Step1: Analyze the molecule diagram
The first molecule diagram is a straight line with three atoms (two terminal, one central), so it's a linear structure. For a linear molecule, the central atom has 2 bonding pairs and 0 lone pairs (since there are no lone pairs shown, and the geometry is linear which corresponds to 2 bonding pairs and 0 lone pairs in VSEPR).
Bonding Pair: 2
Lone Pairs: 0
VSEPR Shape/Geometry: Linear
Second Row (Trigonal Planar):
The molecule diagram for trigonal planar (with 3 bonding pairs and 0 lone pairs) would be a central atom with three terminal atoms forming a triangle (planar) around it. So we can draw a central atom with three atoms attached, each at 120° to each other.
Third Row (Bent or V - shaped with 1 lone pair):
The molecule diagram has a central atom with two terminal atoms and one lone pair (represented by the two dots). The VSEPR shape here is bent (or angular) because of the 1 lone pair. So VSEPR Shape/Geometry: Bent (or Angular)
Fourth Row (Tetrahedral - like with 4 bonding pairs):
The molecule diagram has a central atom with four terminal atoms, which is a tetrahedral geometry. In tetrahedral, the central atom has 4 bonding pairs and 0 lone pairs (since all electron pairs are bonding, as there are no lone pairs shown).
Bonding Pair: 4
Lone Pairs: 0
VSEPR Shape/Geometry: Tetrahedral
Fifth Row (Trigonal Pyramidal):
The molecule diagram has a central atom with three terminal atoms and one lone pair (represented by the two dots). The VSEPR shape here is trigonal pyramidal (because of the 1 lone pair, the shape is a pyramid with a triangular base).
VSEPR Shape/Geometry: Trigonal Pyramidal
The VSEPR angle here is less than 109.5° (since lone pairs repel more than bonding pairs, compressing the angle between the bonding pairs).
Sixth Row (Bent or V - shaped with 2 lone pairs):
The molecule has 2 bonding pairs and 2 lone pairs. The VSEPR shape here is bent (or angular) with a tetrahedral electron - pair geometry but molecular geometry is bent. The molecule diagram would have a central atom with two terminal atoms and two lone pairs (represented by dots).
VSEPR Shape/Geometry: Bent (or Angular)
Now, filling in the table:
| Molecule Diagram (include lone pairs) | # of bonding pairs and lone pairs on central atom | VSEPR Shape/Geometry | VSEPR Angle |
|---|---|---|---|
| <img src="https://via.placeholder.com/80x40?text=Trigonal+Planar" /> (trigonal planar, 3 bonding, 0 lone) | Bonding Pair: 3<br>Lone Pairs: 0 | Trigonal planar | $120^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+1+Lone+Pair" /> (bent, 2 bonding, 1 lone) | Bonding Pair: 2<br>Lone Pairs: 1 | Bent (Angular) | $<120^{\circ}$ |
| <img src="https://via.placeholder.com/60x40?text=Tetrahedral" /> (tetrahedral, 4 bonding, 0 lone) | Bonding Pair: 4<br>Lone Pairs: 0 | Tetrahedral | $109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Trigonal+Pyramidal" /> (trigonal pyramidal, 3 bonding, 1 lone) | Bonding Pair: 3<br>Lone Pairs: 1 | Trigonal Pyramidal | $<109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+2+Lone+Pairs" /> (bent, 2 bonding, 2 lone) | Bonding Pair: 2<br>Lone Pairs: 2 | Bent (Angular) | $<109.5^{\circ}$ |
Final Table:
| Molecule Diagram (include lone pairs) | # of bonding pairs and lone pairs on central atom | VSEPR Shape/Geometry | VSEPR Angle |
|---|---|---|---|
| <img src="https://via.placeholder.com/80x40?text=Trigonal+Planar" /> | Bonding Pair: 3<br>Lone Pairs: 0 | Trigonal planar | $120^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+1+Lone+Pair" /> | Bonding Pair: 2<br>Lone Pairs: 1 | Bent (Angular) | $<120^{\circ}$ |
| <img src="https://via.placeholder.com/60x40?text=Tetrahedral" /> | Bonding Pair: 4<br>Lone Pairs: 0 | Tetrahedral | $109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Trigonal+Pyramidal" /> | Bonding Pair: 3<br>Lone Pairs: 1 | Trigonal Pyramidal | $<109.5^{\circ}$ |
| <img src="https://via.placeholder.com/50x30?text=Bent+2+Lone+Pairs" /> | Bonding Pair: 2<br>Lone Pairs: 2 | Bent (Angular) | $<109.5^{\circ}$ |
If we are only focusing on filling the missing "Bonding Pair", "Lone Pairs" and "VSEPR Shape/Geometry" for each row:
- First row:
- Bonding Pair: $\boldsymbol{2}$
- Lone Pairs: $\boldsymbol{0}$
- VSEPR Shape/Geometry: $\boldsymbol{Linear}$
- Second row: (diagram should be a central atom with 3 atoms attached in a triangle)
- Third row:
- VSEPR Shape/Geometry: $\boldsymbol{Bent\ (Angular)}$
- Fourth row:
- Bonding Pair: $\boldsymbol{4}$
- Lone Pairs: $\boldsymbol{0}$
- VSEPR Shape/Geometry: $\boldsymbol{Tetrahedral}$
- Fifth row:
- VSEPR Shape/Geometry: $\boldsymbol{Trigonal\ Pyramidal}$
- Sixth row:
- VSEPR Shape/Geometry: $\boldsymbol{Bent\ (Angular)}$