QUESTION IMAGE
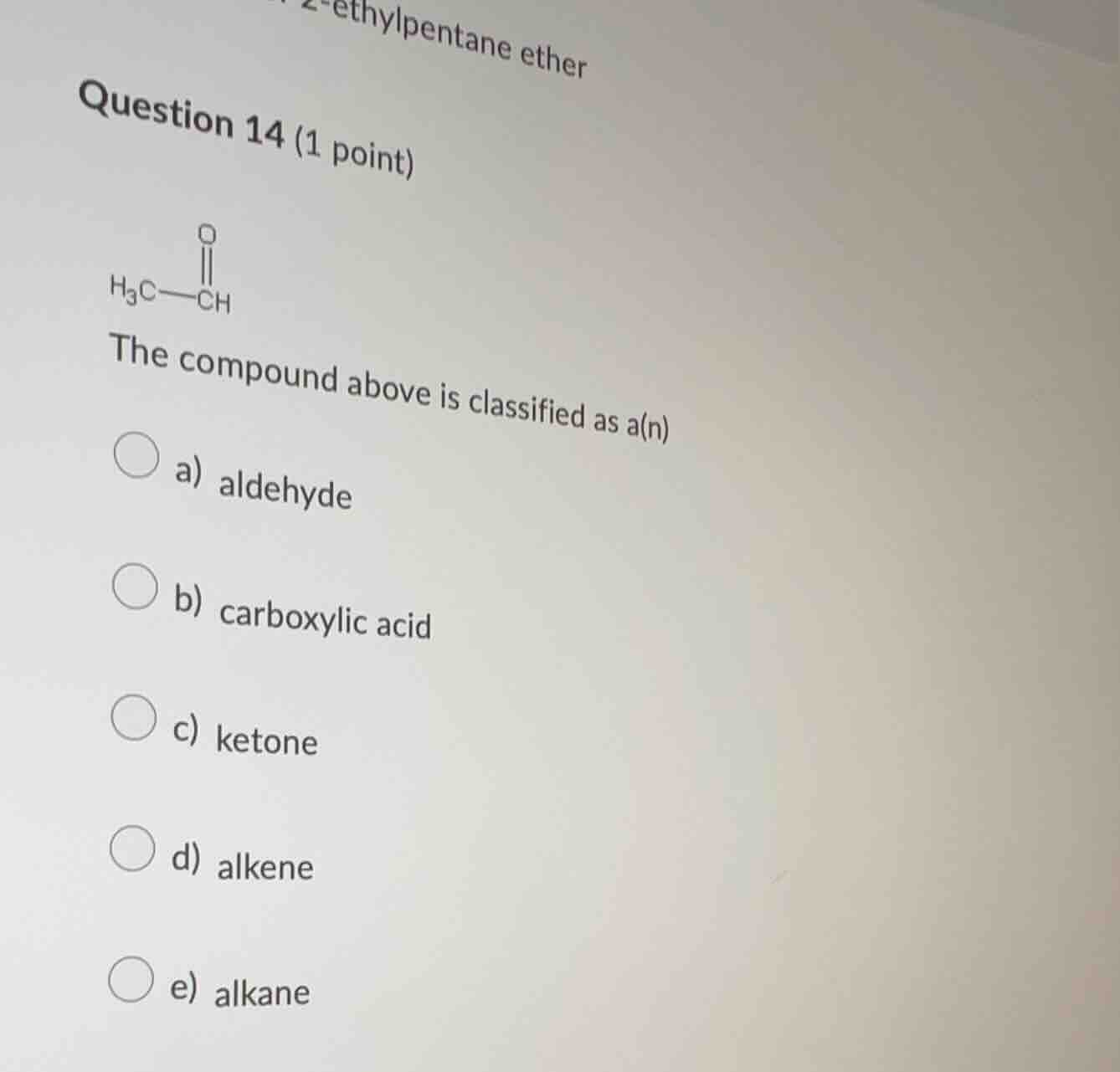
Question
question 14 (1 point)
the compound above is classified as a(n)
a) aldehyde
b) carboxylic acid
c) ketone
d) alkene
e) alkane
Brief Explanations
The given compound has the structure \( \text{H}_3\text{C}-\text{CH}=\text{O} \) (acetaldehyde). Aldehydes have the functional group \( -\text{CHO} \) (carbonyl group with one hydrogen and one carbon attached). Let's analyze other options:
- Carboxylic acids have \( -\text{COOH} \), not present here.
- Ketones have \( \text{C}=\text{O} \) with two carbon groups attached (no hydrogen on the carbonyl carbon), but here the carbonyl carbon is attached to a hydrogen (from \( -\text{CHO} \)) and a methyl group, so it's an aldehyde.
- Alkenes have \( \text{C}=\text{C} \) (not \( \text{C}=\text{O} \)), alkanes have single bonds only (no double bonds), so d and e are out.
- Ketone (c) requires two carbon groups on the carbonyl carbon, which is not the case here (one group is H, one is methyl), so it's an aldehyde.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) aldehyde