QUESTION IMAGE
Question
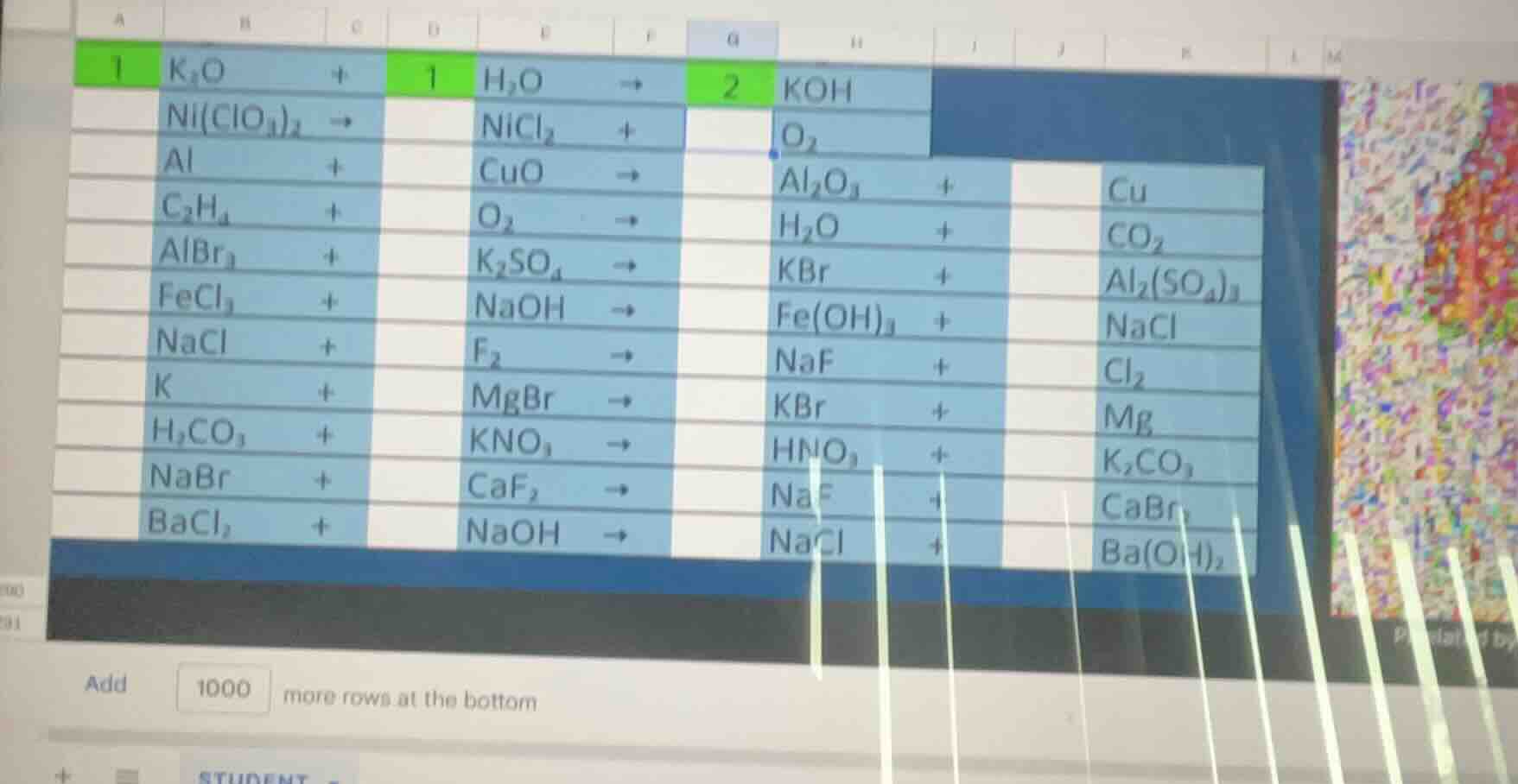
- $ce{k_{2}o + h_{2}o -> koh}$2. $ce{ni(clo_{3})_{2} -> nicl_{2} + o_{2}}$3. $ce{al + cuo -> al_{2}o_{3} + cu}$4. $ce{c_{2}h_{4} + o_{2} -> h_{2}o + co_{2}}$5. $ce{albr_{3} + k_{2}so_{4} -> kbr + al_{2}(so_{4})_{3}}$6. $ce{fecl_{3} + naoh -> fe(oh)_{3} + nacl}$7. $ce{nacl + f_{2} -> naf + cl_{2}}$8. $ce{k + mgbr -> kbr + mg}$9. $ce{h_{2}co_{3} + kno_{3} -> hno_{3} + k_{2}co_{3}}$10. $ce{nabr + caf_{2} -> naf + cabr_{2}}$11. $ce{bacl_{2} + naoh -> nacl + ba(oh)_{2}}$
Step1: Balance $Ni(ClO_3)_2 \to NiCl_2 + O_2$
Count O: 6 on left, 2 on right. Multiply $O_2$ by 3:
$Ni(ClO_3)_2 \to NiCl_2 + 3O_2$
Step2: Balance $Al + CuO \to Al_2O_3 + Cu$
Balance Al: Multiply Al by 2; Balance O: Multiply CuO by 3; Balance Cu: Multiply Cu by 3:
$2Al + 3CuO \to Al_2O_3 + 3Cu$
Step3: Balance $C_2H_4 + O_2 \to H_2O + CO_2$
Balance C: Multiply $CO_2$ by 2; Balance H: Multiply $H_2O$ by 2; Balance O: Multiply $O_2$ by 3:
$C_2H_4 + 3O_2 \to 2H_2O + 2CO_2$
Step4: Balance $AlBr_3 + K_2SO_4 \to KBr + Al_2(SO_4)_3$
Balance Al: Multiply $AlBr_3$ by 2; Balance $SO_4$: Multiply $K_2SO_4$ by 3; Balance K/Br: Multiply $KBr$ by 6:
$2AlBr_3 + 3K_2SO_4 \to 6KBr + Al_2(SO_4)_3$
Step5: Balance $FeCl_3 + NaOH \to Fe(OH)_3 + NaCl$
Balance $OH$: Multiply $NaOH$ by 3; Balance Cl/Na: Multiply $NaCl$ by 3:
$FeCl_3 + 3NaOH \to Fe(OH)_3 + 3NaCl$
Step6: Balance $NaCl + F_2 \to NaF + Cl_2$
Balance Cl: Multiply $NaCl$ by 2; Balance Na/F: Multiply $NaF$ by 2:
$2NaCl + F_2 \to 2NaF + Cl_2$
Step7: Balance $K + MgBr_2 \to KBr + Mg$
Balance Br: Multiply $KBr$ by 2; Balance K: Multiply K by 2:
$2K + MgBr_2 \to 2KBr + Mg$
Step8: Balance $H_2CO_3 + KNO_3 \to HNO_3 + K_2CO_3$
Balance K: Multiply $KNO_3$ by 2; Balance $NO_3$: Multiply $HNO_3$ by 2:
$H_2CO_3 + 2KNO_3 \to 2HNO_3 + K_2CO_3$
Step9: Balance $NaBr + CaF_2 \to NaF + CaBr_2$
Balance Br: Multiply $NaBr$ by 2; Balance Na/F: Multiply $NaF$ by 2:
$2NaBr + CaF_2 \to 2NaF + CaBr_2$
Step10: Balance $BaCl_2 + NaOH \to NaCl + Ba(OH)_2$
Balance $OH$: Multiply $NaOH$ by 2; Balance Na/Cl: Multiply $NaCl$ by 2:
$BaCl_2 + 2NaOH \to 2NaCl + Ba(OH)_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $K_2O + H_2O \to 2KOH$
- $Ni(ClO_3)_2 \to NiCl_2 + 3O_2$
- $2Al + 3CuO \to Al_2O_3 + 3Cu$
- $C_2H_4 + 3O_2 \to 2H_2O + 2CO_2$
- $2AlBr_3 + 3K_2SO_4 \to 6KBr + Al_2(SO_4)_3$
- $FeCl_3 + 3NaOH \to Fe(OH)_3 + 3NaCl$
- $2NaCl + F_2 \to 2NaF + Cl_2$
- $2K + MgBr_2 \to 2KBr + Mg$
- $H_2CO_3 + 2KNO_3 \to 2HNO_3 + K_2CO_3$
- $2NaBr + CaF_2 \to 2NaF + CaBr_2$
- $BaCl_2 + 2NaOH \to 2NaCl + Ba(OH)_2$