QUESTION IMAGE
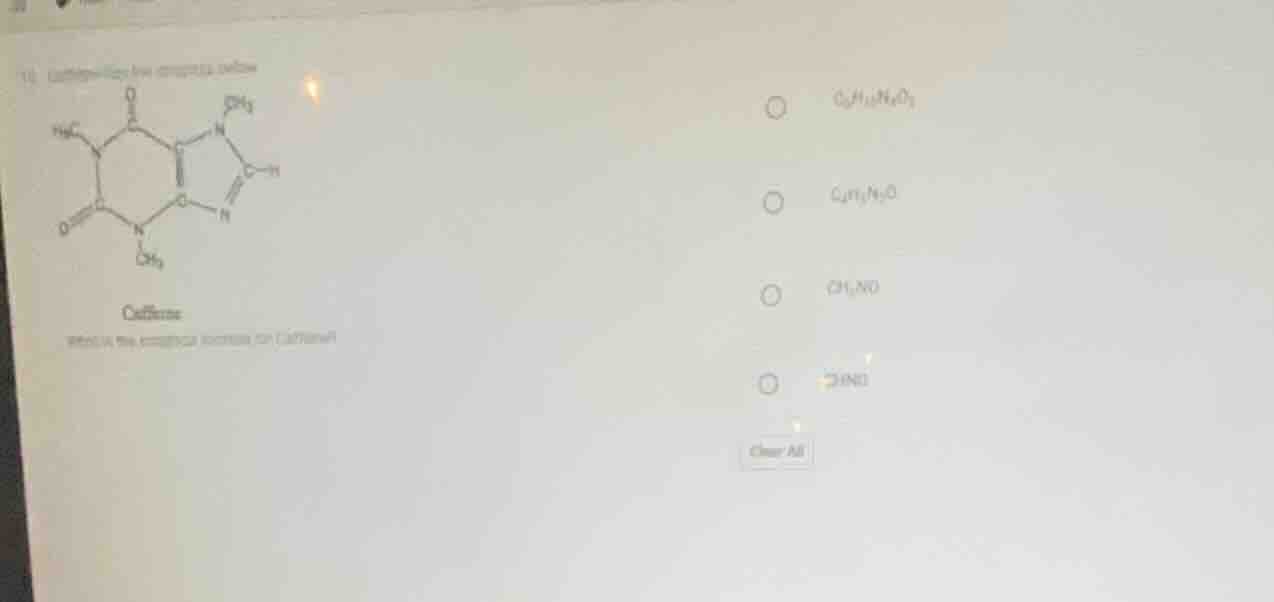
Question
- caffeine... caffeine... choose the correct molecular formula for caffeine? options: \\( \ce{c8h10n4o2} \\), \\( \ce{c4h5n2o} \\), \\( \ce{ch2no} \\), another option with unclear text clear all
Step1: Identify atoms in Caffeine
Caffeine's structure: Count C, H, N, O. From the diagram, Caffeine has 8 C, 10 H, 4 N, 2 O? Wait, no, correct caffeine formula is \( C_8H_{10}N_4O_2 \)? Wait, no, wait the options: Wait the options given: Wait the image shows options, maybe I misread. Wait, let's re-express. Wait, caffeine's molecular formula is \( C_8H_{10}N_4O_2 \)? Wait no, wait the options: Wait the first option is \( C_8H_{10}N_4O_2 \)? Wait the user's image: Let's parse the options. Wait the first option: \( C_8H_{10}N_4O_2 \), second \( C_{10}H_8N_4O \), third \( CH_3NO \), fourth \( C_2H_3NO \). Wait, no, maybe I made a mistake. Wait, caffeine's structure: the ring structure. Let's count: C atoms: 8? Wait, no, standard caffeine formula is \( C_8H_{10}N_4O_2 \). Wait, but let's check the options. Wait the first option is \( C_8H_{10}N_4O_2 \) (assuming the first option's formula is \( C_8H_{10}N_4O_2 \)). Wait, maybe the first option is \( C_8H_{10}N_4O_2 \), which matches caffeine's molecular formula. Wait, let's confirm: Caffeine has 8 carbon, 10 hydrogen, 4 nitrogen, 2 oxygen atoms. So the correct formula is \( C_8H_{10}N_4O_2 \), which should be the first option.
Step2: Match with options
Among the options, the first option (assuming it's \( C_8H_{10}N_4O_2 \)) is the molecular formula of caffeine.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \boldsymbol{C_8H_{10}N_4O_2} \) (the first option, e.g., A. \( C_8H_{10}N_4O_2 \) if options are labeled A, B, C, D)