QUESTION IMAGE
Question
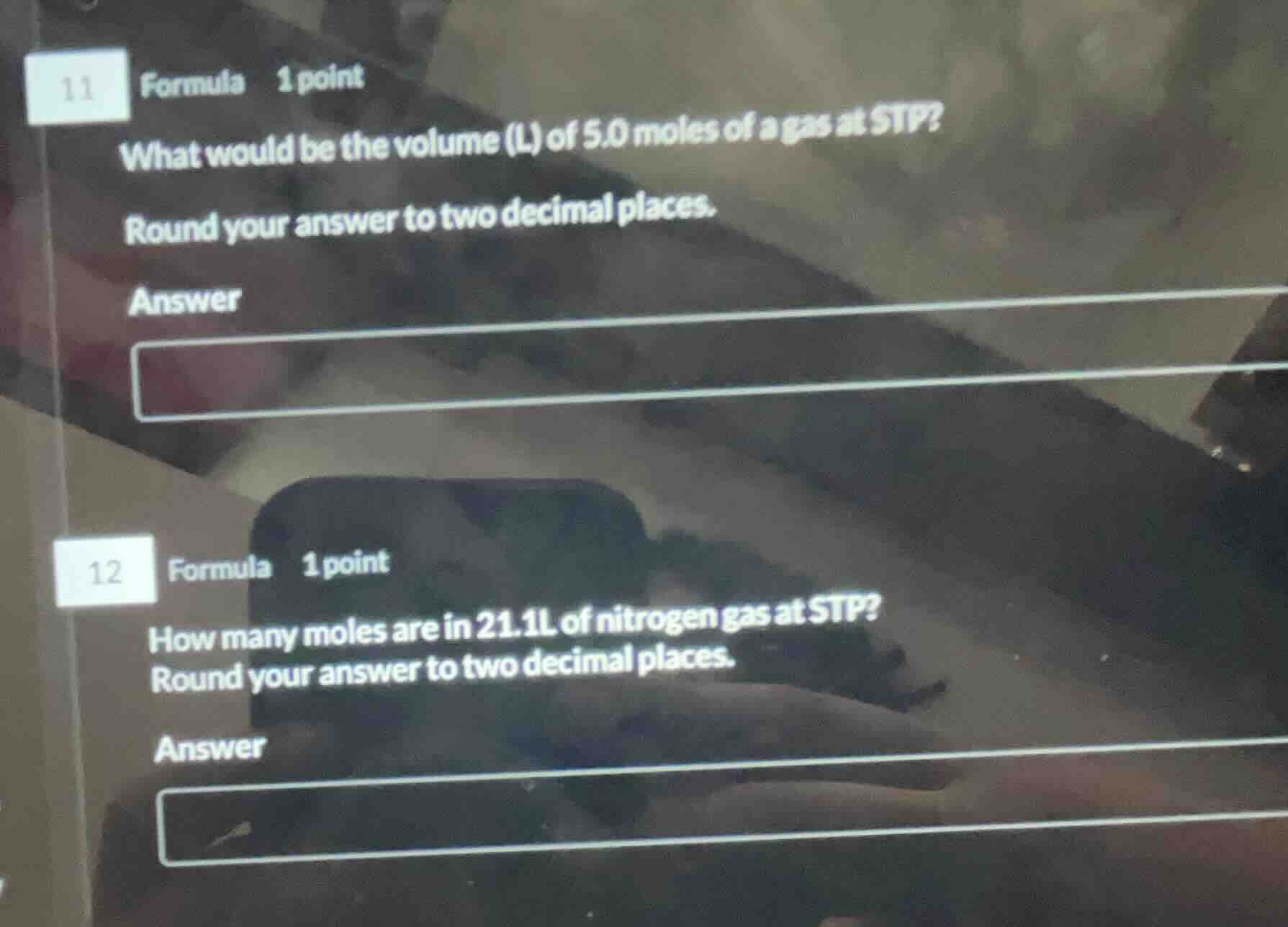
11 formula 1 point
what would be the volume (l) of 5.0 moles of a gas at stp?
round your answer to two decimal places.
answer
12 formula 1 point
how many moles are in 21.1l of nitrogen gas at stp?
round your answer to two decimal places.
answer
Step1: Recall STP molar volume
At STP, 1 mole of gas = 22.4 L
Step2: Calculate volume for Q11
$\text{Volume} = 5.0 \times 22.4 = 112.00$ L
Step3: Calculate moles for Q12
$\text{Moles} = \frac{21.1}{22.4} \approx 0.94$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 112.00 L
- 0.94 moles