QUESTION IMAGE
Question
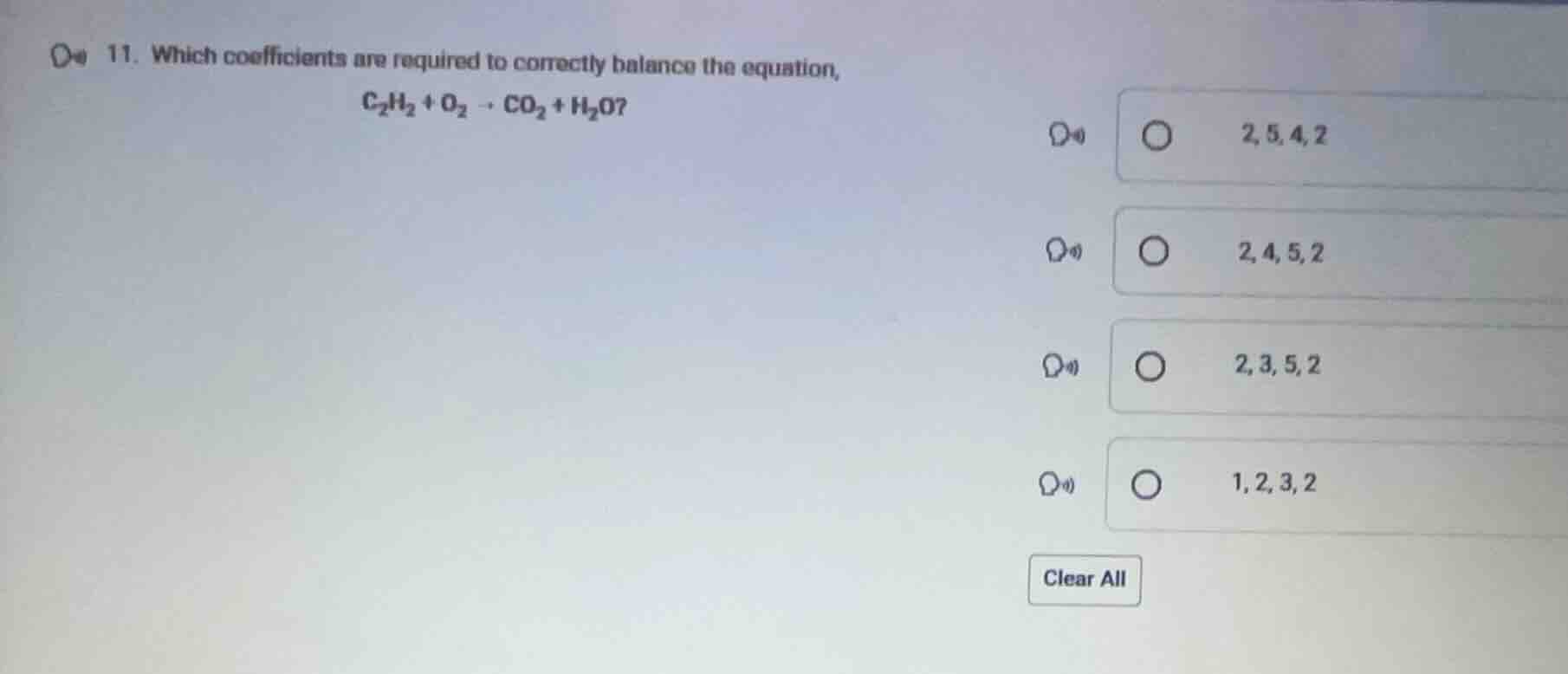
- which coefficients are required to correctly balance the equation, \\(\ce{c_{2}h_{2} + o_{2} -> co_{2} + h_{2}o}?\\) \\(\boldsymbol{2,5,4,2}\\) \\(\boldsymbol{2,4,5,2}\\) \\(\boldsymbol{2,3,5,2}\\) \\(\boldsymbol{1,2,3,2}\\) clear all
Step1: Balance Carbon atoms
In \( C_2H_2 \), there are 2 C atoms. So, we put a coefficient of 2 in front of \( CO_2 \) to balance C: \( C_2H_2 + O_2
ightarrow 2CO_2 + H_2O \)
Step2: Balance Hydrogen atoms
In \( C_2H_2 \), there are 2 H atoms. In \( H_2O \), to balance H, we need 1 \( H_2O \) (but wait, original \( C_2H_2 \) has 2 H, so actually, if we have 2 \( C_2H_2 \), then H atoms: 2*2 = 4. So we put 2 in front of \( H_2O \): \( 2C_2H_2 + O_2
ightarrow 2CO_2 + 2H_2O \)? Wait, no, let's start over. Let's let the coefficient of \( C_2H_2 \) be \( x \), \( O_2 \) be \( y \), \( CO_2 \) be \( z \), \( H_2O \) be \( w \). So:
- C: \( 2x = z \)
- H: \( 2x = 2w \) (so \( x = w \))
- O: \( 2y = 2z + w \)
Let's take \( x = 2 \) (to make z even, since O is diatomic). Then z = 4 (from 2x = z, x=2, z=4). w = x = 2. Then O: 2y = 2*4 + 2 = 10, so y = 5. So the equation is \( 2C_2H_2 + 5O_2
ightarrow 4CO_2 + 2H_2O \). So coefficients are 2,5,4,2.
Step3: Verify the first option
Check the first option: 2 (for \( C_2H_2 \)), 5 (for \( O_2 \)), 4 (for \( CO_2 \)), 2 (for \( H_2O \)). Let's count atoms:
- C: 22 = 4; 41 = 4 (balanced)
- H: 22 = 4; 22 = 4 (balanced)
- O: 52 = 10; 42 + 2*1 = 8 + 2 = 10 (balanced)
So this works.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2, 5, 4, 2 (the first option, e.g., A. 2, 5, 4, 2 if options are labeled A,B,C,D with first as A)