QUESTION IMAGE
Question
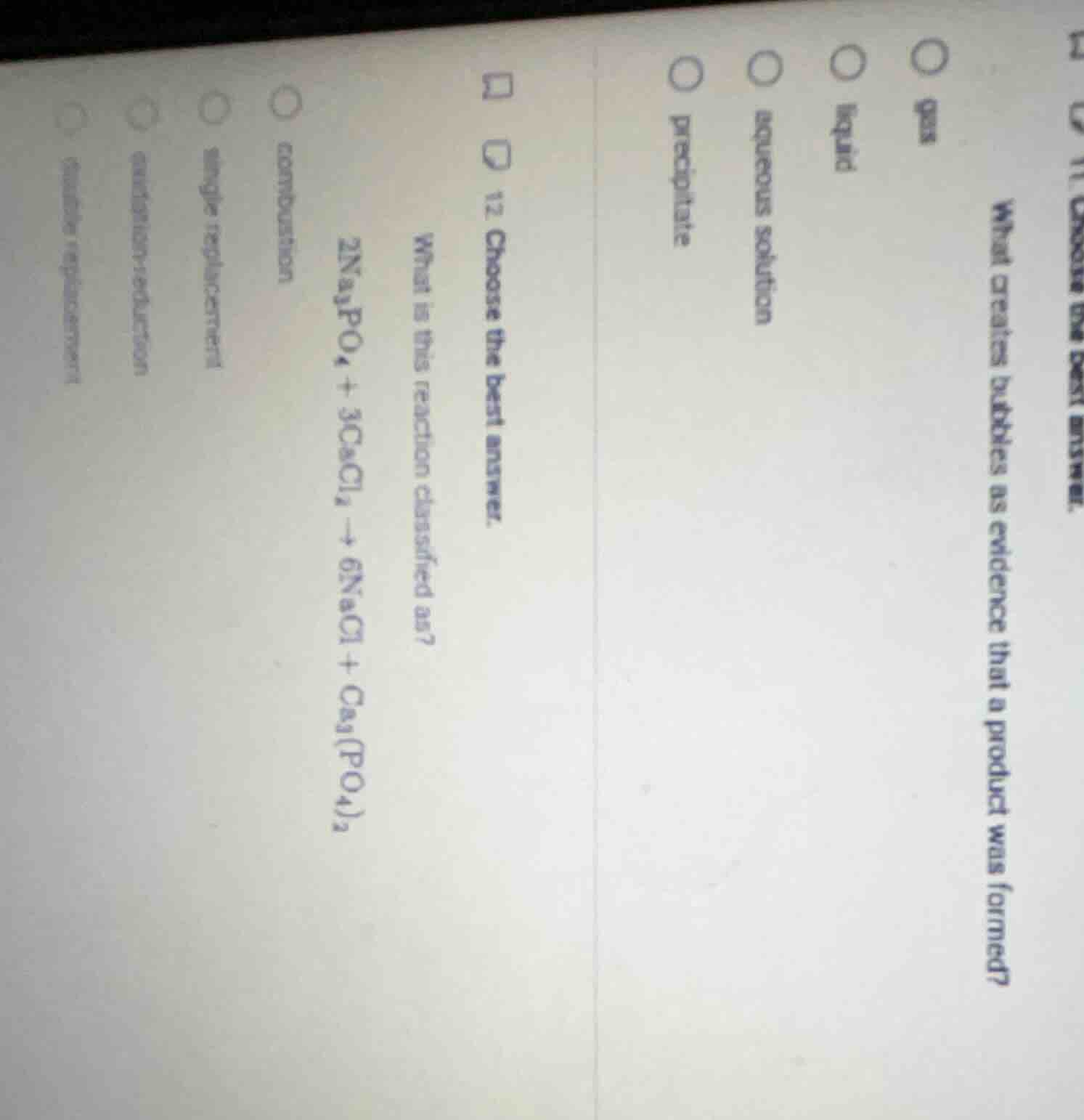
- choose the best answer. what is this reaction classified as? $2na_3po_4 + 3cacl_2 \
ightarrow 6nacl + ca_3(po_4)_2$ combustion single replacement oxidation - reduction double replacement
Brief Explanations
- For question 11: Bubbles are visual evidence that a gaseous product has formed, as gases disperse from a reaction mixture in this way.
- For question 12: The reaction $2Na_3PO_4 + 3CaCl_2
ightarrow 6NaCl + Ca_3(PO_4)_2$ involves two ionic compounds swapping their positive and negative ions to form new compounds, which is a double replacement reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- gas
- double replacement