QUESTION IMAGE
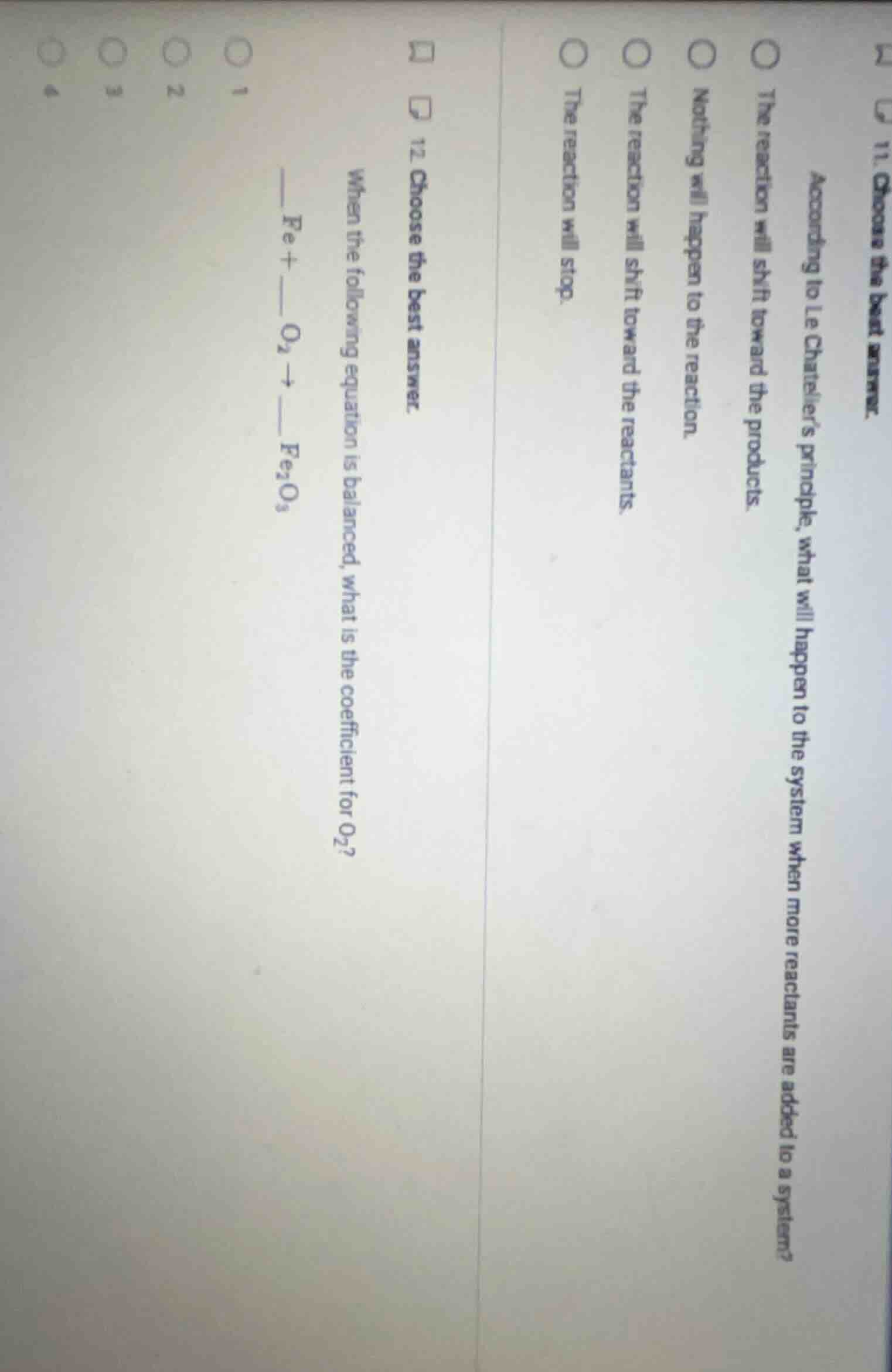
Question
- choose the best answer. when the following equation is balanced, what is the coefficient for o₂? _ fe + _ o₂ → ___ fe₂o₃ 1 2 3 4
Step1: Solve Q11 via Le Chatelier's Principle
Le Chatelier's principle states that adding reactants drives equilibrium to consume excess reactants by forming more products.
Step2: Balance Fe in Q12
Match Fe atoms on both sides: $4\text{Fe} + \_\_\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$
Step3: Balance O in Q12
Match O atoms on both sides: $4\text{Fe} + 3\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The reaction will shift toward the products.
- 3