QUESTION IMAGE
Question
- part 1
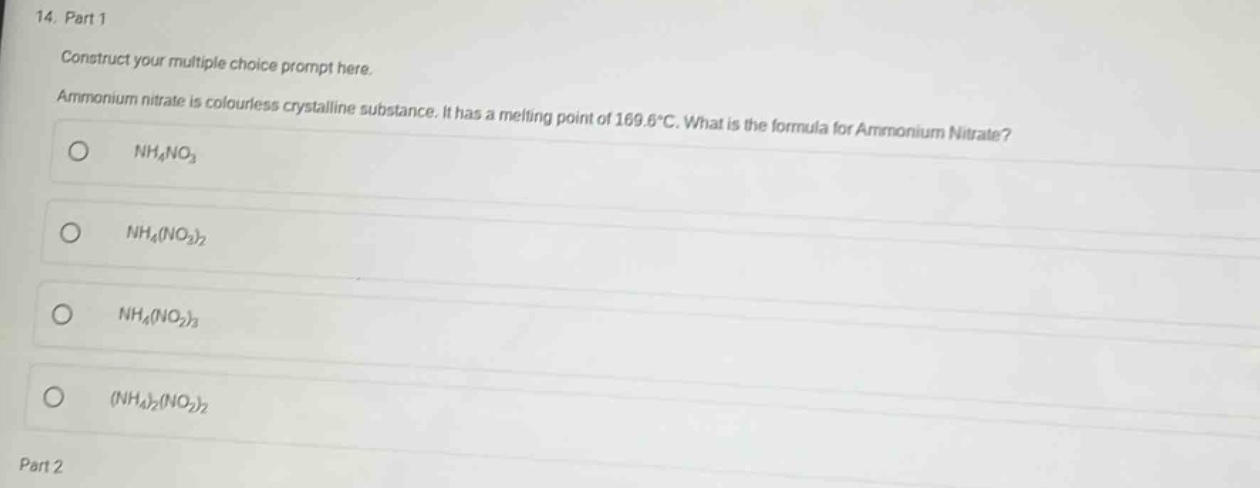
construct your multiple choice prompt here.
ammonium nitrate is colourless crystalline substance. it has a melting point of 169.6°c. what is the formula for ammonium nitrate?
- $nh_4no_3$
- $nh_4(no_2)_2$
- $nh_4(no_2)_3$
- $(nh_4)_2(no_2)_2$
part 2
To determine the formula for ammonium nitrate, we analyze the ions involved. Ammonium ion is \(NH_4^+\) and nitrate ion is \(NO_3^-\). For a neutral compound, the charges must balance. The charge of \(NH_4^+\) is +1 and \(NO_3^-\) is -1, so one of each ion combines to form \(NH_4NO_3\). The other options have incorrect combinations of ions (e.g., \(NO_2^-\) is nitrite, not nitrate, and the subscripts are wrong for balancing charges).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(NH_4NO_3\)