QUESTION IMAGE
Question
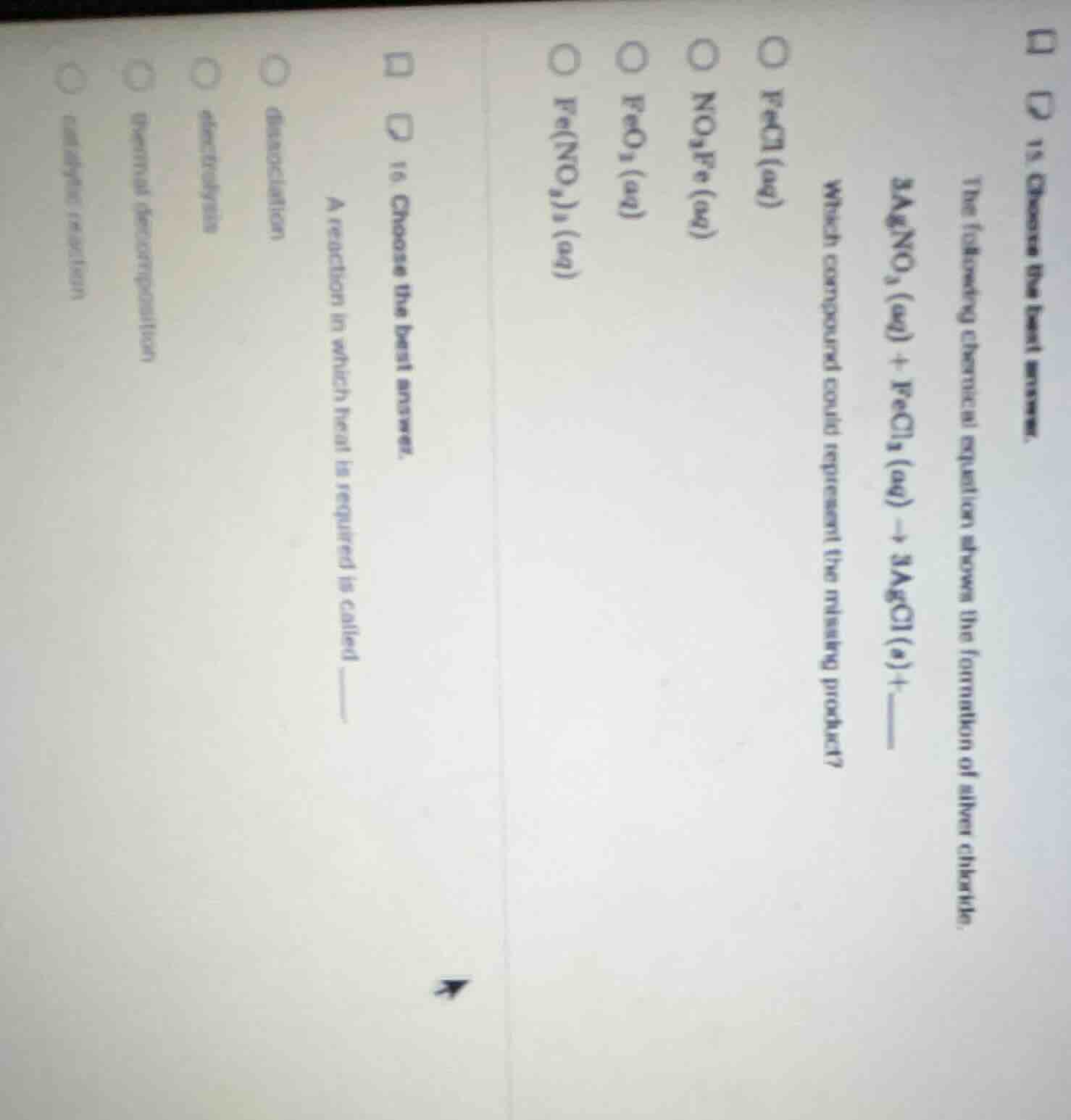
- choose the best answer. the following chemical equation shows the formation of silver chloride. 3agno₃(aq) + fecl₃(aq) → 3agcl(s) + which compound could represent the missing product? fecl(aq) no₃fe(aq) feo₃(aq) fe(no₃)₃(aq) 16. choose the best answer. a reaction in which heat is required is called... dissociation electrolysis thermal decomposition endothermic reaction
Step1: Balance cations/anions for double displacement
The reaction is a double displacement: $\text{AgNO}_3(aq) + \text{FeCl}_3(aq)
ightarrow \text{AgCl}(s) + ?$.
$\text{Fe}^{3+}$ from $\text{FeCl}_3$ combines with $\text{NO}_3^-$ from $\text{AgNO}_3$.
Step2: Write balanced missing product
Combine $\text{Fe}^{3+}$ and $3\text{NO}_3^-$ to form $\text{Fe(NO}_3\text{)}_3(aq)$.
Balanced full equation: $3\text{AgNO}_3(aq) + \text{FeCl}_3(aq)
ightarrow 3\text{AgCl}(s) + \text{Fe(NO}_3\text{)}_3(aq)$
Step3: Identify heat-required reaction definition
A reaction needing heat input for decomposition is thermal decomposition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{Fe(NO}_3\text{)}_3(aq)$
- thermal decomposition