QUESTION IMAGE
Question
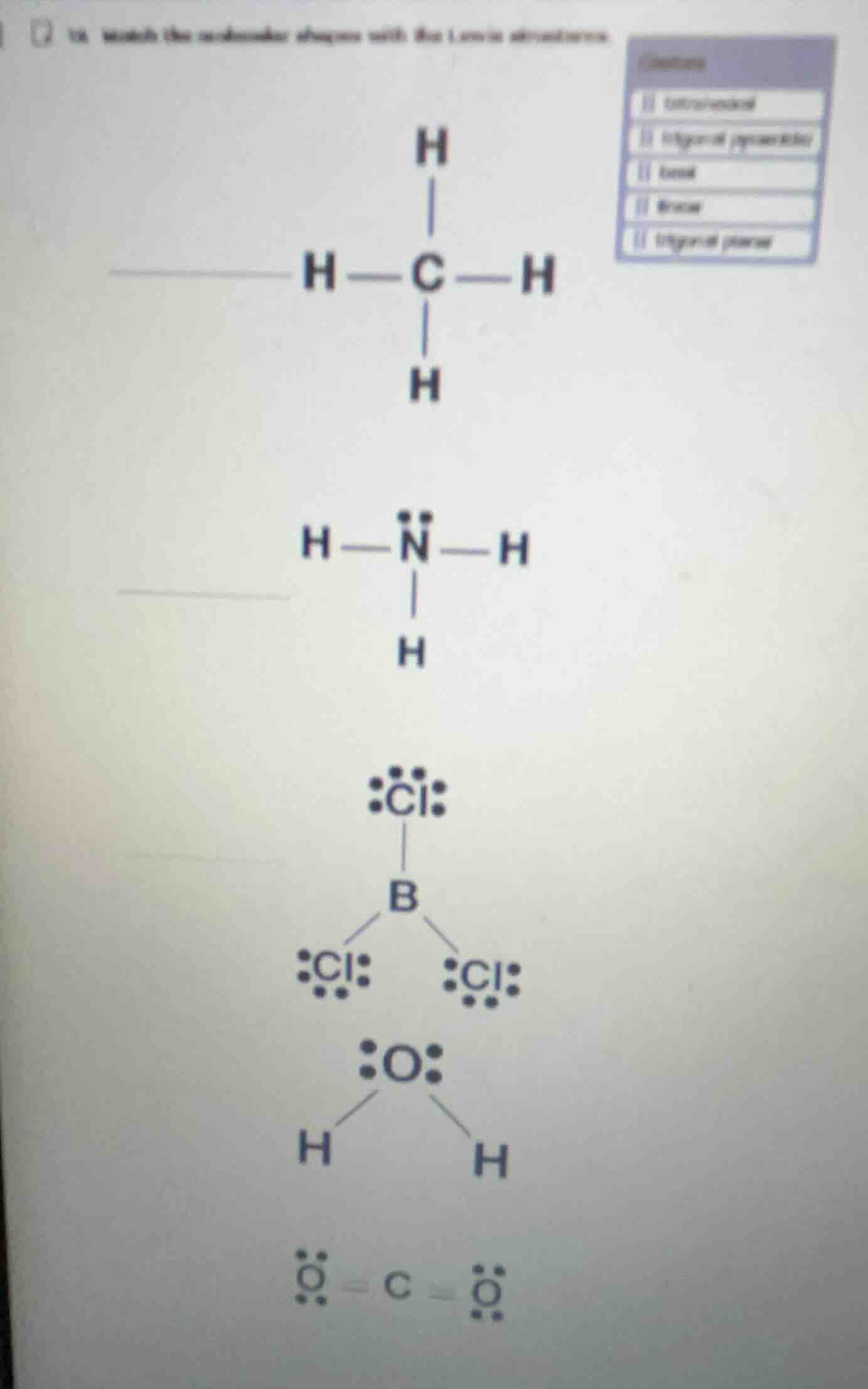
- match the molecule diagrams with the lewis structures (the right side has a list of options, and the left side has several molecular structures: the first is the tetrahedral structure of ch₄, the second is the pyramidal structure of nh₃ with a lone pair on n, the third is the trigonal planar structure of bcl₃, the fourth is the bent structure of h₂o with two lone pairs on o, the fifth is the linear structure of co₂ with double bonds and lone pairs on o)
Brief Explanations
- CH₄ (Methane): The central C has 4 bonding pairs, 0 lone pairs. Electron domain geometry is tetrahedral, matching the label "tetrahedral".
- NH₃ (Ammonia): The central N has 3 bonding pairs, 1 lone pair. Electron domain geometry is tetrahedral, but molecular geometry is trigonal pyramidal, matching "trigonal pyramidal".
- BCl₃ (Boron Trichloride): The central B has 3 bonding pairs, 0 lone pairs. Both electron and molecular geometry are trigonal planar, matching "trigonal planar".
- H₂O (Water): The central O has 2 bonding pairs, 2 lone pairs. Electron domain geometry is tetrahedral, molecular geometry is bent, matching "bent".
- CO₂ (Carbon Dioxide): The central C has 2 double bonding pairs, 0 lone pairs. Both electron and molecular geometry are linear, matching "linear".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{H-\underset{\underset{H}{|}}{C}-H}$ (CH₄) → Tetrahedral
- $\boldsymbol{H-\underset{\underset{H}{|}}{\overset{\overset{..}{N}}{}}-H}$ (NH₃) → Trigonal pyramidal
- $\boldsymbol{:\underset{\underset{..}{Cl}}{Cl}-B-\underset{..}{\overset{..}{Cl}}:}$ (BCl₃) → Trigonal planar
- $\boldsymbol{H-\underset{..}{\overset{..}{O}}-H}$ (H₂O) → Bent
- $\boldsymbol{:\underset{..}{\overset{..}{O}}=C=\underset{..}{\overset{..}{O}}:}$ (CO₂) → Linear