QUESTION IMAGE
Question
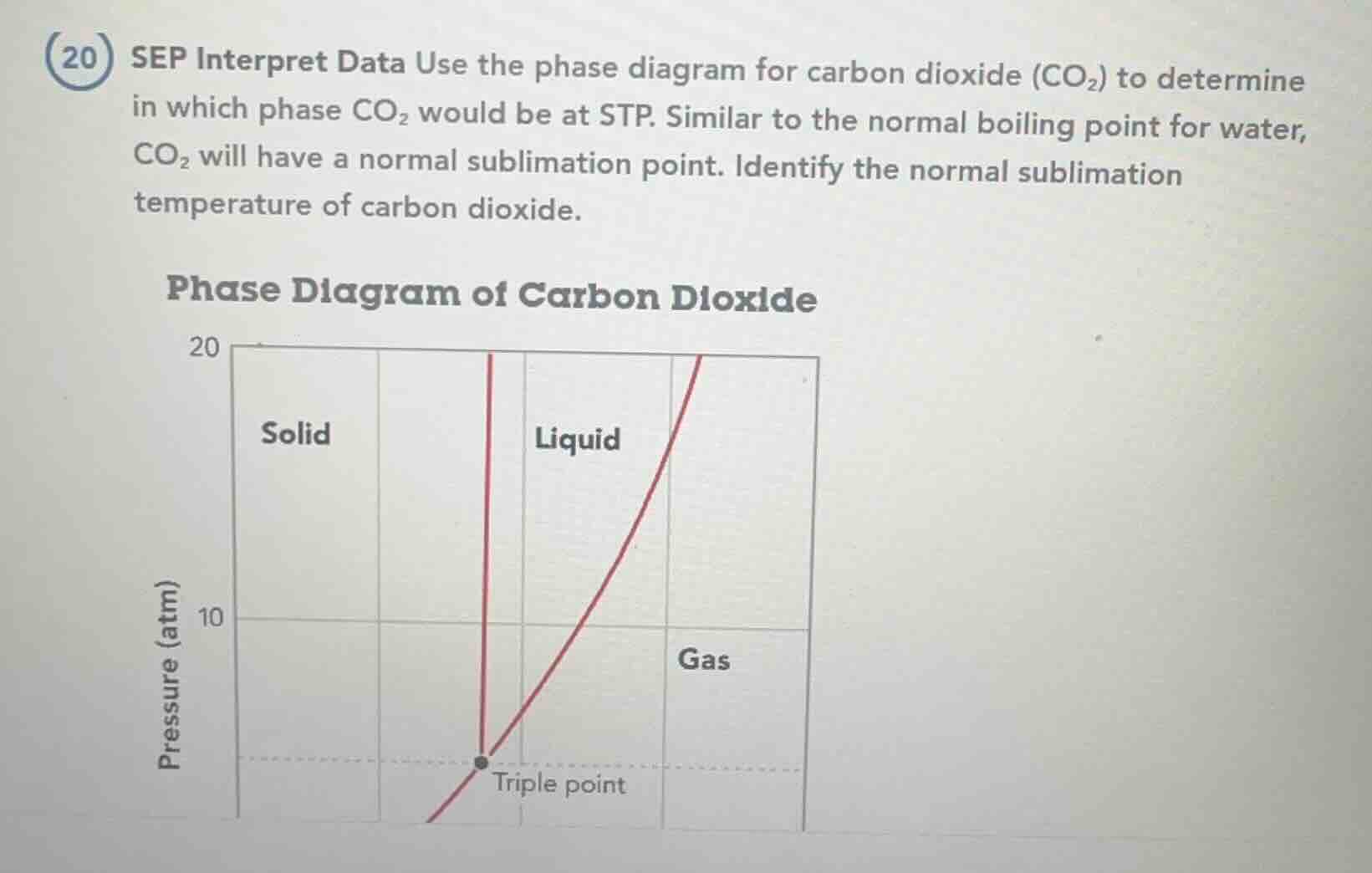
(20) sep interpret data use the phase diagram for carbon dioxide (co₂) to determine in which phase co₂ would be at stp. similar to the normal boiling point for water, co₂ will have a normal sublimation point. identify the normal sublimation temperature of carbon dioxide. phase diagram of carbon dioxide
Step1: Define STP conditions
STP = 1 atm, 273.15 K (0°C)
Step2: Locate STP on phase diagram
The triple point of CO₂ is ~5.18 atm, -56.6°C. Since 1 atm < triple point pressure, at 0°C and 1 atm, CO₂ lies in the gas region.
Step3: Define normal sublimation point
Normal sublimation occurs at 1 atm, where solid directly transitions to gas. This is the temperature where the solid-gas boundary meets 1 atm.
Step4: Identify sublimation temperature
From standard CO₂ phase diagram data, this temperature is -78.5°C (194.7 K).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- At STP, CO₂ is in the gas phase.
- The normal sublimation temperature of carbon dioxide is $\boldsymbol{-78.5^\circ C}$ (or 194.7 K).