QUESTION IMAGE
Question
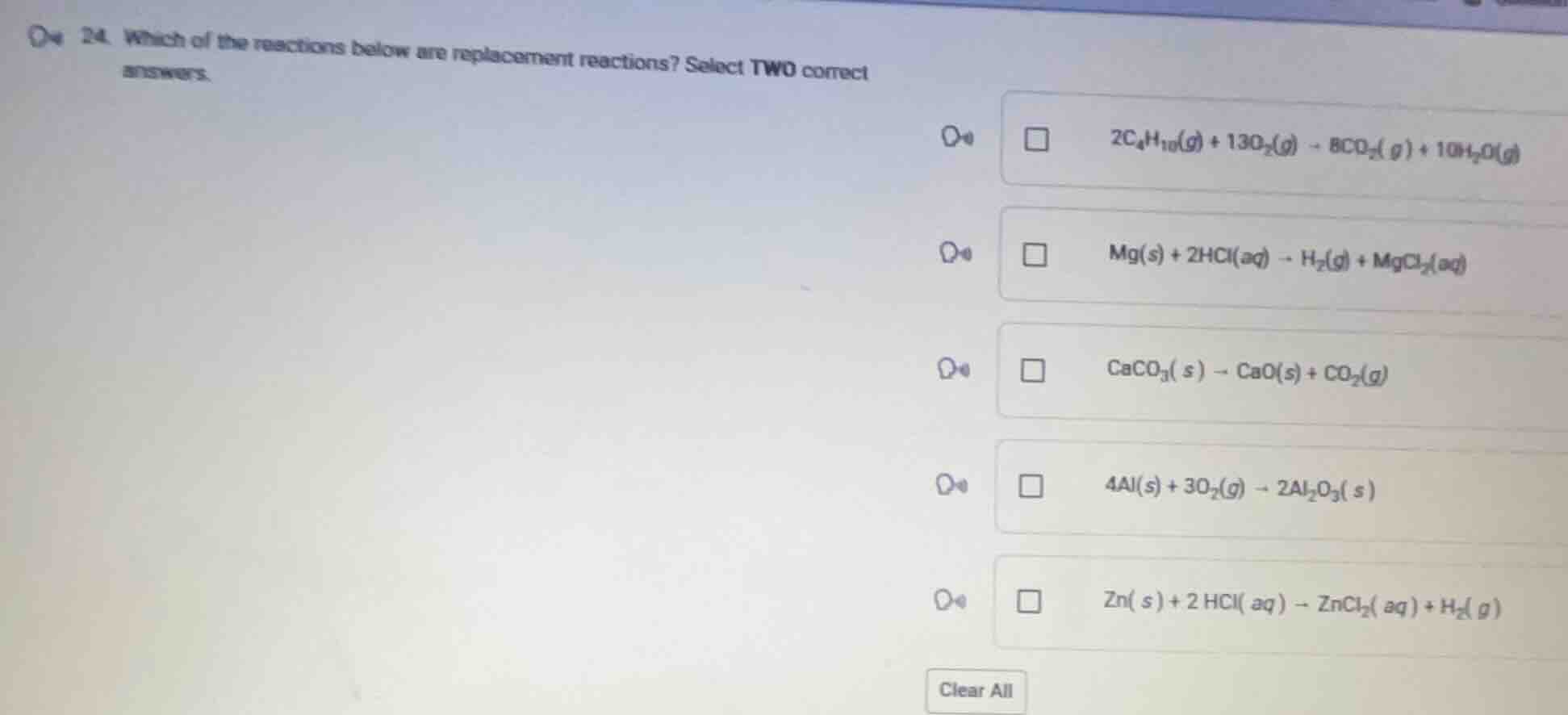
- which of the reactions below are replacement reactions? select two correct answers.
$2c_4h_{10}(g) + 13o_2(g) \
ightarrow 8co_2(g) + 10h_2o(g)$
$mg(s) + 2hcl(aq) \
ightarrow h_2(g) + mgcl_2(aq)$
$caco_3(s) \
ightarrow cao(s) + co_2(g)$
$4al(s) + 3o_2(g) \
ightarrow 2al_2o_3(s)$
$zn(s) + 2hcl(aq) \
ightarrow zncl_2(aq) + h_2(g)$
Step1: Define replacement reactions
A single replacement reaction follows the general form: $\text{A} + \text{BC}
ightarrow \text{AC} + \text{B}$, where an element replaces another in a compound.
Step2: Analyze Option A
This is a combustion reaction: $\text{2C}_4\text{H}_{10}(g) + 13\text{O}_2(g)
ightarrow 8\text{CO}_2(g) + 10\text{H}_2\text{O}(g)$. It fits $\text{C}_x\text{H}_y + \text{O}_2
ightarrow \text{CO}_2 + \text{H}_2\text{O}$, not replacement.
Step3: Analyze Option B
Fits single replacement: $\text{Mg}(s) + 2\text{HCl}(aq)
ightarrow \text{H}_2(g) + \text{MgCl}_2(aq)$. Mg replaces H in HCl.
Step4: Analyze Option C
This is a decomposition reaction: $\text{CaCO}_3(s)
ightarrow \text{CaO}(s) + \text{CO}_2(g)$. It fits $\text{AB}
ightarrow \text{A} + \text{B}$, not replacement.
Step5: Analyze Option D
This is a synthesis reaction: $\text{4Al}(s) + 3\text{O}_2(g)
ightarrow 2\text{Al}_2\text{O}_3(s)$. It fits $\text{A} + \text{B}
ightarrow \text{AB}$, not replacement.
Step6: Analyze Option E
Fits single replacement: $\text{Zn}(s) + 2\text{HCl}(aq)
ightarrow \text{ZnCl}_2(aq) + \text{H}_2(g)$. Zn replaces H in HCl.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{Mg}(s) + 2\text{HCl}(aq)
ightarrow \text{H}_2(g) + \text{MgCl}_2(aq)$
E. $\text{Zn}(s) + 2\text{HCl}(aq)
ightarrow \text{ZnCl}_2(aq) + \text{H}_2(g)$