QUESTION IMAGE
Question
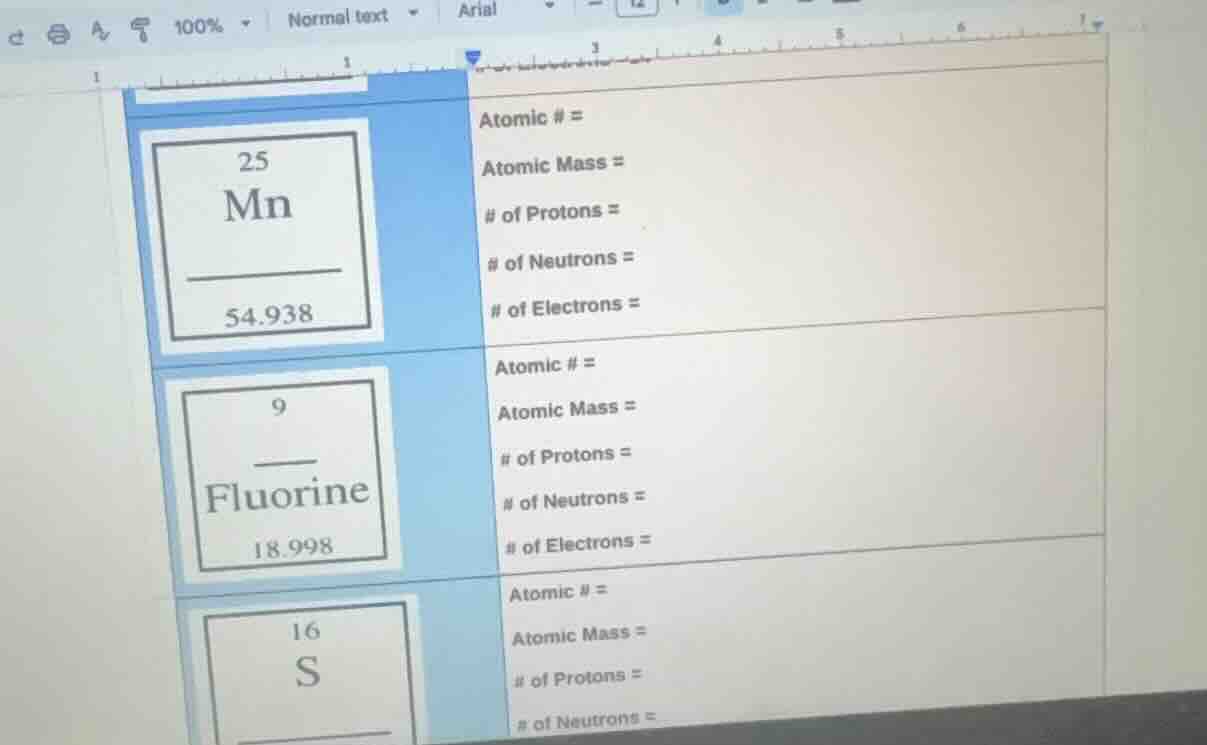
25
mn
54.938
atomic # =
atomic mass =
of protons =
of neutrons =
of electrons =
9
fluorine
18.998
atomic # =
atomic mass =
of protons =
of neutrons =
of electrons =
16
s
atomic # =
atomic mass =
of protons =
of neutrons =
For Manganese (Mn):
Step1: Atomic Number
The atomic number is the top number on the element tile, so for Mn, Atomic \# = 25.
Step2: Atomic Mass
The atomic mass is the bottom number, so Atomic Mass = 54.938.
Step3: Number of Protons
Protons = Atomic Number, so \# of Protons = 25.
Step4: Number of Neutrons
Neutrons = Atomic Mass (rounded) - Atomic Number. Rounded atomic mass of Mn is 55, so Neutrons = 55 - 25 = 30.
Step5: Number of Electrons
Electrons = Protons (in neutral atom), so \# of Electrons = 25.
For Fluorine (F):
Step1: Atomic Number
Atomic \# = 9 (top number).
Step2: Atomic Mass
Atomic Mass = 18.998 (bottom number).
Step3: Number of Protons
\# of Protons = 9 (same as atomic number).
Step4: Number of Neutrons
Rounded atomic mass is 19, so Neutrons = 19 - 9 = 10.
Step5: Number of Electrons
\# of Electrons = 9 (neutral atom).
For Sulfur (S):
Step1: Atomic Number
Atomic \# = 16 (top number).
Step2: Atomic Mass
We know sulfur's atomic mass is approximately 32.06 (commonly, or from the tile's blank, but using known value).
Step3: Number of Protons
\# of Protons = 16 (atomic number).
Step4: Number of Neutrons
Neutrons = 32 - 16 = 16 (using rounded atomic mass 32).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (filled for each element):
Manganese (Mn):
Atomic \# = 25
Atomic Mass = 54.938
\# of Protons = 25
\# of Neutrons = 30
\# of Electrons = 25
Fluorine (F):
Atomic \# = 9
Atomic Mass = 18.998
\# of Protons = 9
\# of Neutrons = 10
\# of Electrons = 9
Sulfur (S):
Atomic \# = 16
Atomic Mass = 32.06 (or as per tile's blank, but standard is ~32.06)
\# of Protons = 16
\# of Neutrons = 16 (using atomic mass 32)
\# of Electrons = 16 (neutral atom)
(Note: For sulfur's atomic mass, if using the tile's blank, we can also calculate from neutrons, but standard atomic mass is 32.06. The blank for sulfur's atomic mass (bottom) would be ~32.06, and neutrons = 32 - 16 = 16.)