QUESTION IMAGE
Question
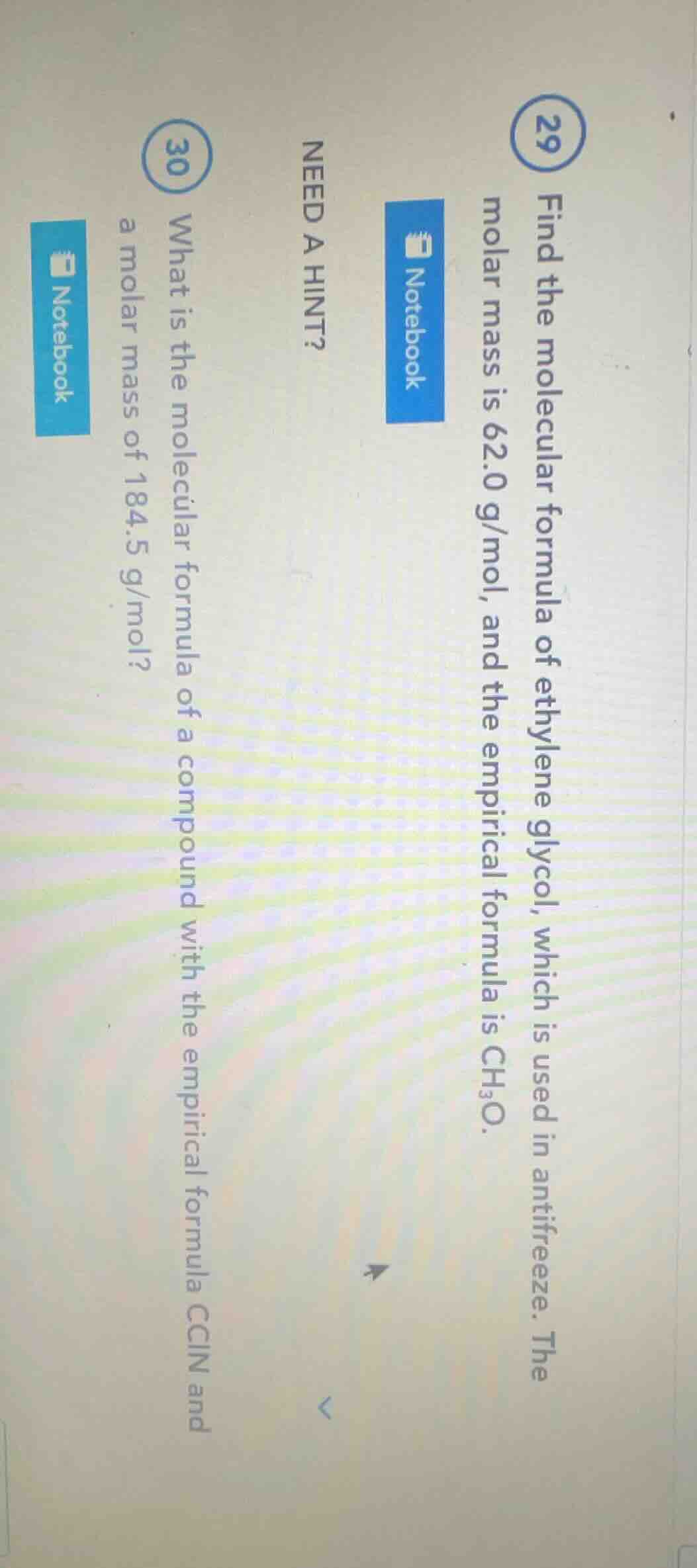
29 find the molecular formula of ethylene glycol, which is used in antifreeze. the molar mass is 62.0 g/mol, and the empirical formula is ch₃o. need a hint? 30 what is the molecular formula of a compound with the empirical formula ccln and a molar mass of 184.5 g/mol?
Step1: Calculate empirical formula mass (CH₃O)
Find sum of atomic masses: $M_{\text{empirical}} = 12.01 + 3(1.008) + 16.00 = 31.034\ \text{g/mol}$
Step2: Find ratio n of molar to empirical mass
$n = \frac{\text{Molar Mass}}{\text{Empirical Formula Mass}} = \frac{62.0}{31.034} \approx 2$
Step3: Multiply empirical formula by n
Molecular formula: $2 \times \text{CH}_3\text{O} = \text{C}_2\text{H}_6\text{O}_2$
---
Step1: Calculate empirical formula mass (CCIN)
Find sum of atomic masses: $M_{\text{empirical}} = 12.01 + 12.01 + 126.90 + 14.01 = 164.93\ \text{g/mol}$
Step2: Find ratio n of molar to empirical mass
$n = \frac{\text{Molar Mass}}{\text{Empirical Formula Mass}} = \frac{184.5}{164.93} \approx 1.12$ (round to 1, as n must be integer)
Step3: Multiply empirical formula by n
Molecular formula: $1 \times \text{CCIN} = \text{C}_2\text{ClN}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{C}_2\text{H}_6\text{O}_2$
- $\text{C}_2\text{ClN}$