QUESTION IMAGE
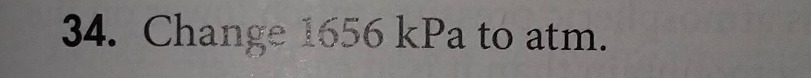
Question
- change 1656 kpa to atm.
Step1: Recall the conversion factor
We know that \(1\ \text{atm} = 101.325\ \text{kPa}\). So to convert from kPa to atm, we use the formula: \(\text{Pressure in atm}=\frac{\text{Pressure in kPa}}{101.325}\)
Step2: Substitute the given value
Given pressure is \(1656\ \text{kPa}\). Substitute into the formula: \(\text{Pressure in atm}=\frac{1656}{101.325}\)
Step3: Calculate the result
\(\frac{1656}{101.325}\approx16.34\) (rounded to two decimal places)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(1656\ \text{kPa}\) is approximately \(16.34\ \text{atm}\)