QUESTION IMAGE
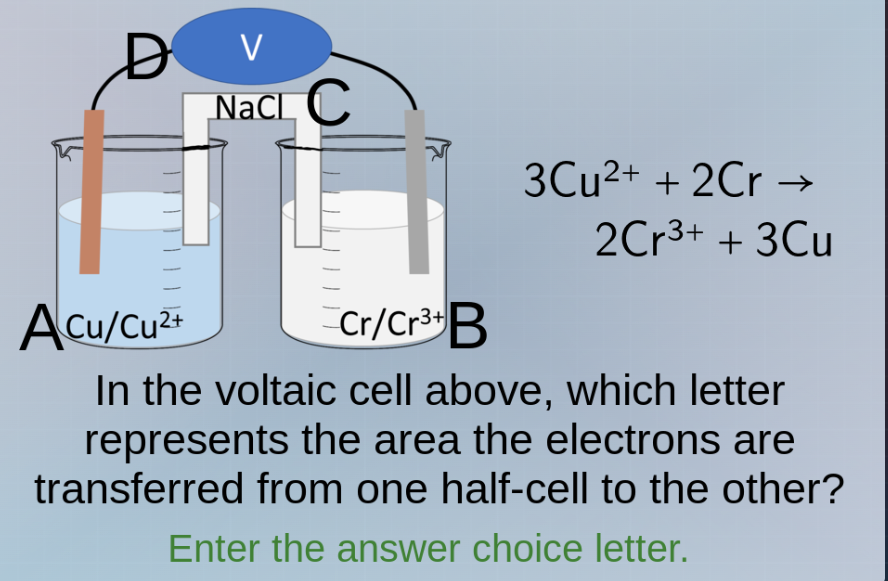
Question
3cu²⁺ + 2cr → 2cr³⁺ + 3cu in the voltaic cell above, which letter represents the area the electrons are transferred from one half - cell to the other? enter the answer choice letter.
In a voltaic cell, electrons flow through the external wire (the part labeled V, with connections D and C). The anode (where oxidation occurs, here Cr is oxidized: \( \text{Cr}
ightarrow \text{Cr}^{3+} + 3e^- \)) releases electrons, and these electrons travel through the external circuit (the wire, which includes the area with the voltmeter V, connected by D) to the cathode (where reduction occurs, \( \text{Cu}^{2+} + 2e^-
ightarrow \text{Cu} \)). So the area where electrons are transferred (through the external wire) is represented by D (or the wire part, but among the letters, D is the connection in the external circuit for electron flow). Wait, let's recheck: the half-cells are A (Cu/Cu²⁺) and B (Cr/Cr³⁺). The external circuit is the wire with V, connecting D (from A's electrode) and C (from B's electrode). Electrons flow from the anode (B, Cr is anode as it's oxidized) to cathode (A, Cu is cathode as Cu²⁺ is reduced) through the external wire. So the path of electrons is from B's electrode (Cr) through C, V, D to A's electrode (Cu). So the area (the wire part) where electrons are transferred is the external circuit, which is the part with D (the wire connecting the two electrodes via the voltmeter). Wait, the question is which letter represents the area electrons are transferred from one half - cell to the other. The transfer is through the external wire, which is the part with D (the wire segment) or the voltmeter's connection. But looking at the labels: D is the wire from A's electrode, C is from B's electrode, V is the voltmeter. The electrons move from B (anode) to A (cathode) through the external circuit (C - V - D). So the area (the wire path) is represented by D (or the external circuit, which is the path including D). Wait, maybe the answer is D. Wait, let's confirm the anode and cathode: oxidation at anode (Cr → Cr³⁺ + 3e⁻), so anode is B (Cr electrode). Reduction at cathode (Cu²⁺ + 2e⁻ → Cu), so cathode is A (Cu electrode). Electrons flow from anode (B) to cathode (A) through the external wire (C - V - D). So the area where electrons are transferred (the external wire) is represented by D (the wire connecting to the cathode) or the path. But the letters are D, C, A, B. So the external circuit (where electrons move) is the wire with V, connecting D (from A) and C (from B). So the transfer happens through the external wire, which is the part with D (or the wire segment). So the answer is D? Wait, maybe I made a mistake. Wait, the question is "which letter represents the area the electrons are transferred from one half - cell to the other". The two half - cells are A and B. The transfer between them is through the external circuit (the wire with V, D, and C). So the letter for the external circuit's electron transfer path is D (the wire from A's electrode) or C (from B's electrode)? Wait, electrons go from B (anode) to A (cathode), so from B's electrode (Cr) through C, then V, then D to A's electrode (Cu). So the path is C - V - D. So the area (the wire) where electrons are transferred is the external circuit, which is represented by D (the wire connecting to the cathode) or C (connecting to the anode). But the question is which letter. Let's see the diagram: D is connected to A's electrode, C to B's. The electrons move from C (anode side) through V to D (cathode side). So the area (the wire path) is the external circuit, which is the part with D (the wire segment) or C. Wait, maybe the answer is D. Alternatively, maybe the answer is D. Let's check again: the electrons are transferred from the anode half - cell (B) to the…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D