QUESTION IMAGE
Question
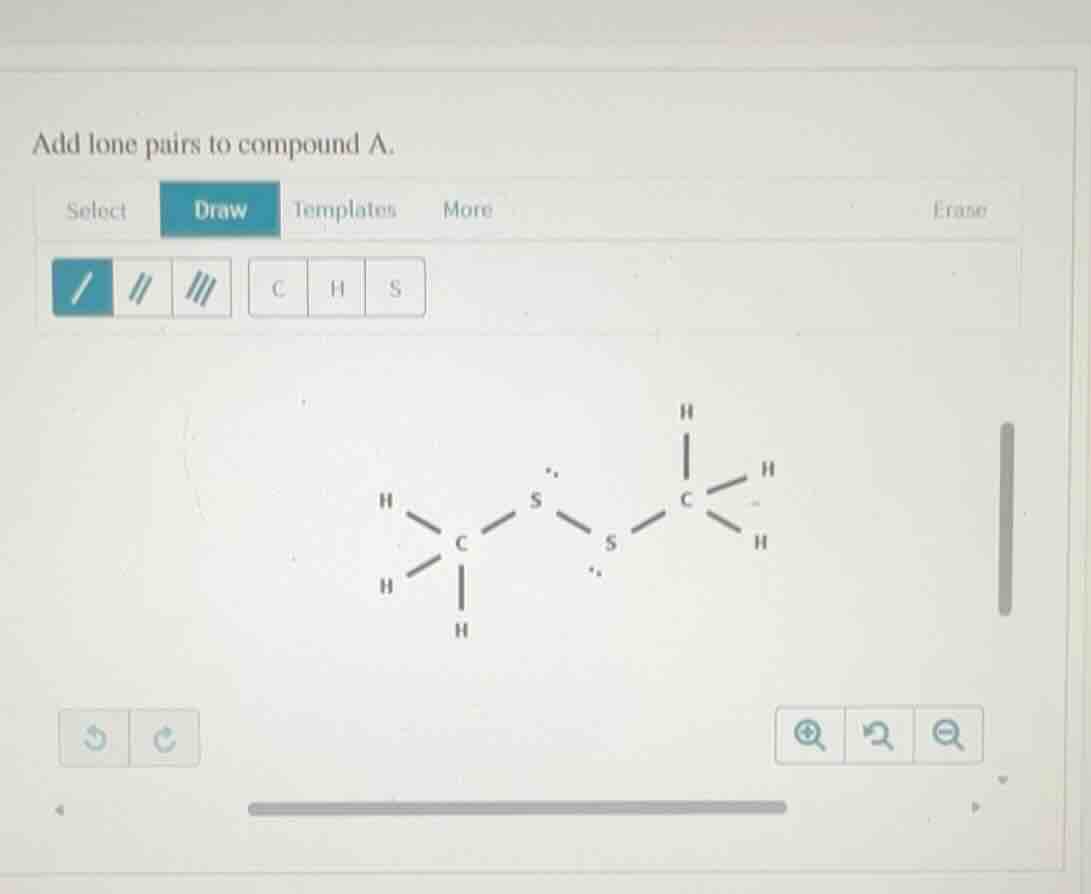
add lone pairs to compound a.
Step1: Determine valence electrons of S
Sulfur has 6 valence electrons.
Step2: Count bonded electrons per S
Each S forms 2 single bonds, so uses 4 electrons.
Step3: Calculate lone pair electrons per S
Lone pair electrons = $6 - 4 = 2$ pairs (4 total electrons).
Step4: Add missing lone pairs
Each S needs 2 lone pairs; the given structure has 1 pair per S, so add 1 more pair to each S.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The final structure has each sulfur atom with 2 lone pairs:
H
|
H - C - S::S::C - H
| |
H H(Each :: represents 2 lone pair electrons, so each S has 4 non-bonding electrons in two lone pairs)