QUESTION IMAGE
Question
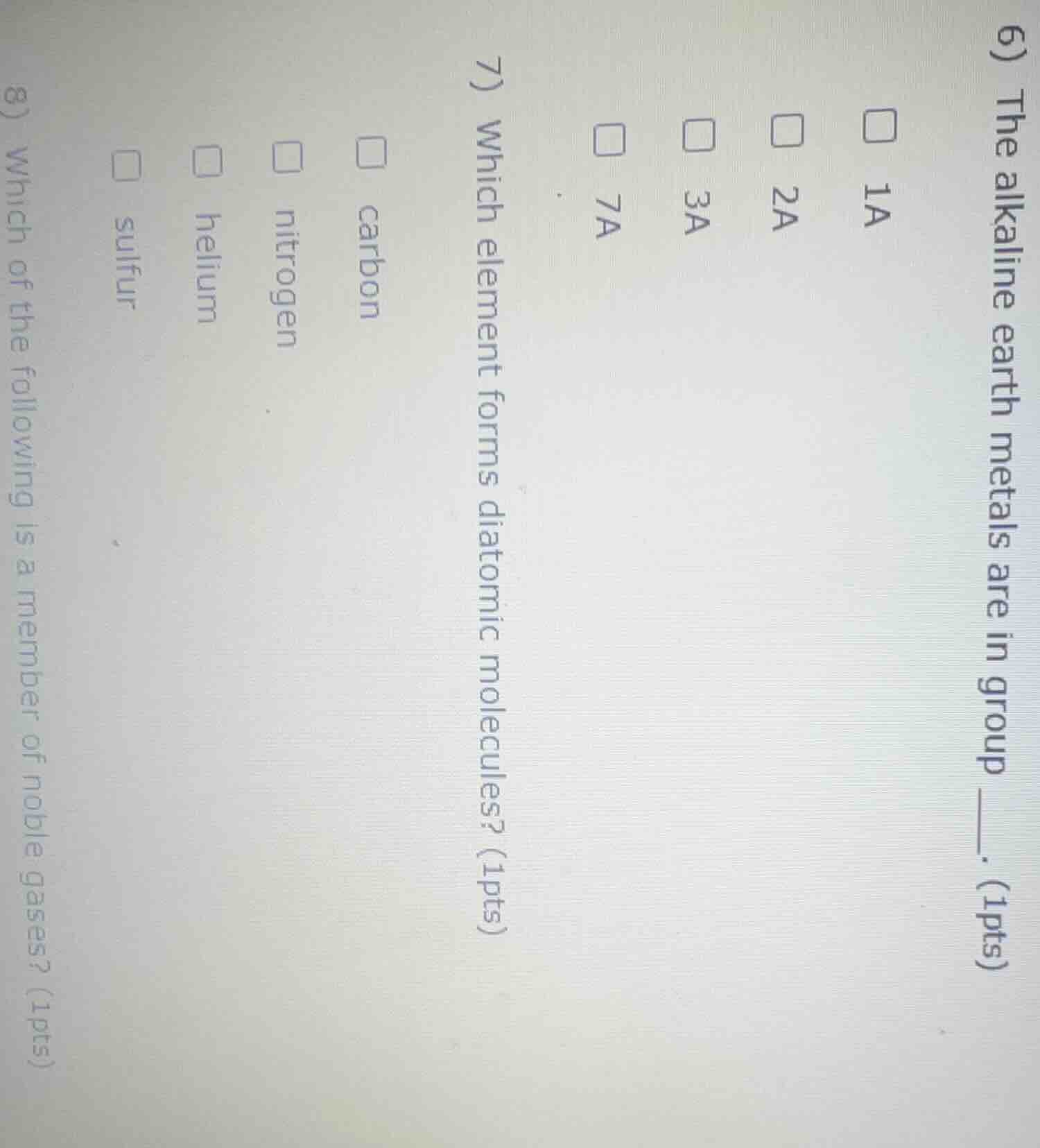
- the alkaline earth metals are in group ____. (1pts)
□ 1a
□ 2a
□ 3a
□ 7a
- which element forms diatomic molecules? (1pts)
□ carbon
□ nitrogen
□ helium
□ sulfur
- which of the following is a member of noble gases? (1pts)
Brief Explanations
- Alkaline earth metals are defined as the group 2A (or group 2) elements in the periodic table, which include beryllium, magnesium, calcium, strontium, barium, and radium.
- Diatomic molecules consist of two atoms bonded together. Nitrogen naturally exists as $\text{N}_2$, a diatomic molecule; carbon, helium, and sulfur do not form stable diatomic molecules under standard conditions.
- Noble gases are the group 18 elements, which include helium, neon, argon, krypton, xenon, and radon. Carbon, nitrogen, and sulfur are not noble gases.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 2A
- nitrogen
- helium