QUESTION IMAGE
Question
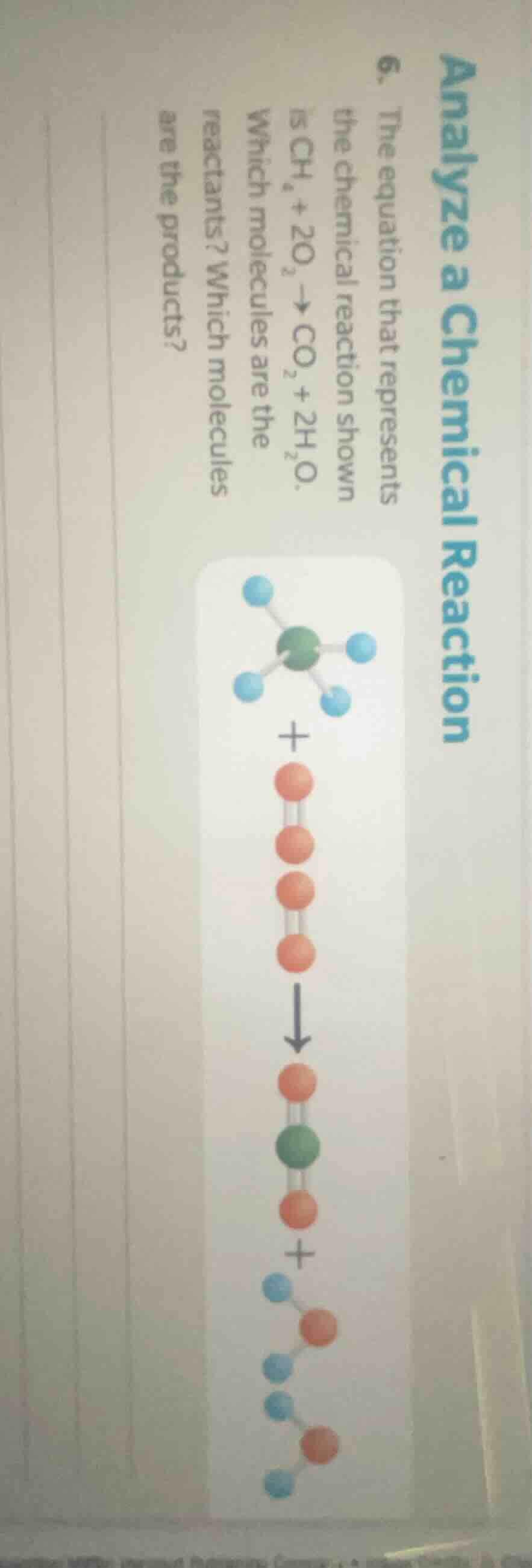
analyze a chemical reaction
- the equation that represents
the chemical reaction shown
is $\ce{ch_{4} + 2o_{2} \
ightarrow co_{2} + 2h_{2}o}$.
which molecules are the
reactants? which molecules
are the products?
(there is a chemical reaction diagram in the original question, which is omitted here)
In a chemical reaction, reactants are the substances that undergo the reaction (on the left side of the arrow), and products are the substances formed (on the right side of the arrow). For the equation $\ce{CH_{4} + 2O_{2} -> CO_{2} + 2H_{2}O}$, we identify reactants and products based on their position relative to the arrow.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Reactants: $\ce{CH_{4}}$ (methane) and $\ce{O_{2}}$ (oxygen) molecules.
- Products: $\ce{CO_{2}}$ (carbon dioxide) and $\ce{H_{2}O}$ (water) molecules.