QUESTION IMAGE
Question
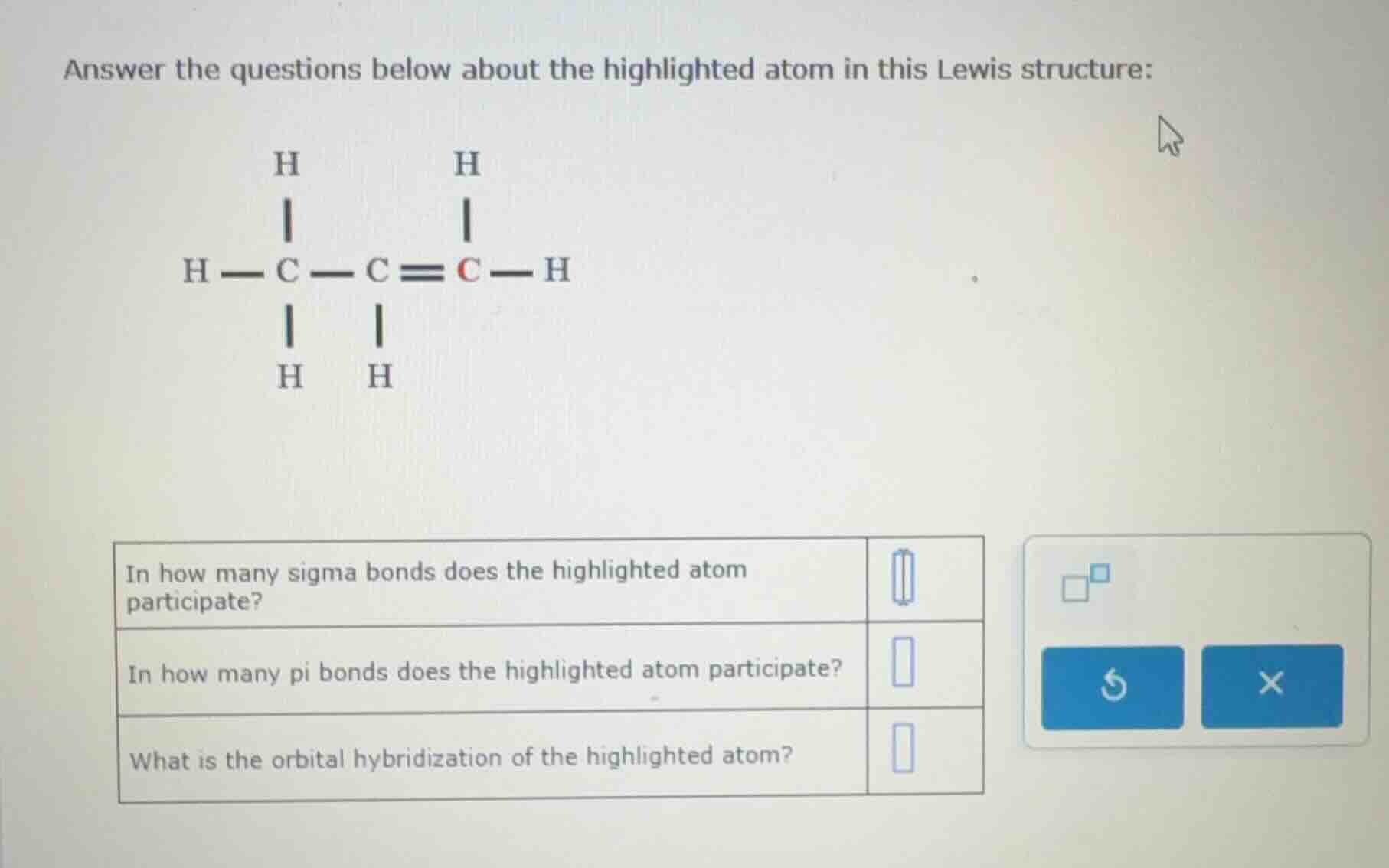
answer the questions below about the highlighted atom in this lewis structure:
h
|
h — c — c ≡ c — h
|
h
h
|
h
|
h
|
in how many sigma bonds does the highlighted atom participate?
in how many pi bonds does the highlighted atom participate?
what is the orbital hybridization of the highlighted atom?
Part 1: Sigma Bonds
Step1: Identify bonds of highlighted C
The highlighted C is bonded to one C (triple bond) and one H (single bond). A triple bond has 1 sigma, single bond has 1 sigma.
Step2: Count sigma bonds
From C - C triple bond: 1 sigma. From C - H single bond: 1 sigma. Total sigma bonds: \(1 + 1 = 2\).
Part 2: Pi Bonds
Step1: Analyze pi bonds in triple bond
A triple bond has 2 pi bonds (since triple bond = 1 sigma + 2 pi). The highlighted C is in a triple bond with another C.
Step2: Count pi bonds
So pi bonds: 2.
Part 3: Hybridization
Step1: Determine bonding type
The highlighted C has a triple bond (1 sigma, 2 pi) and one single bond (1 sigma), total 2 sigma bonds. For carbon, \(sp\) hybridization occurs when there are 2 sigma bonds (linear geometry, bond angle \(180^\circ\)), as \(sp\) hybrid orbitals form 2 sigma bonds, and unhybridized p - orbitals form pi bonds.
Step2: Conclude hybridization
So hybridization is \(sp\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- Sigma bonds: \(\boldsymbol{2}\)
- Pi bonds: \(\boldsymbol{2}\)
- Hybridization: \(\boldsymbol{sp}\)