QUESTION IMAGE
Question
anthocyanin experiment
choosing the experimental wavelength
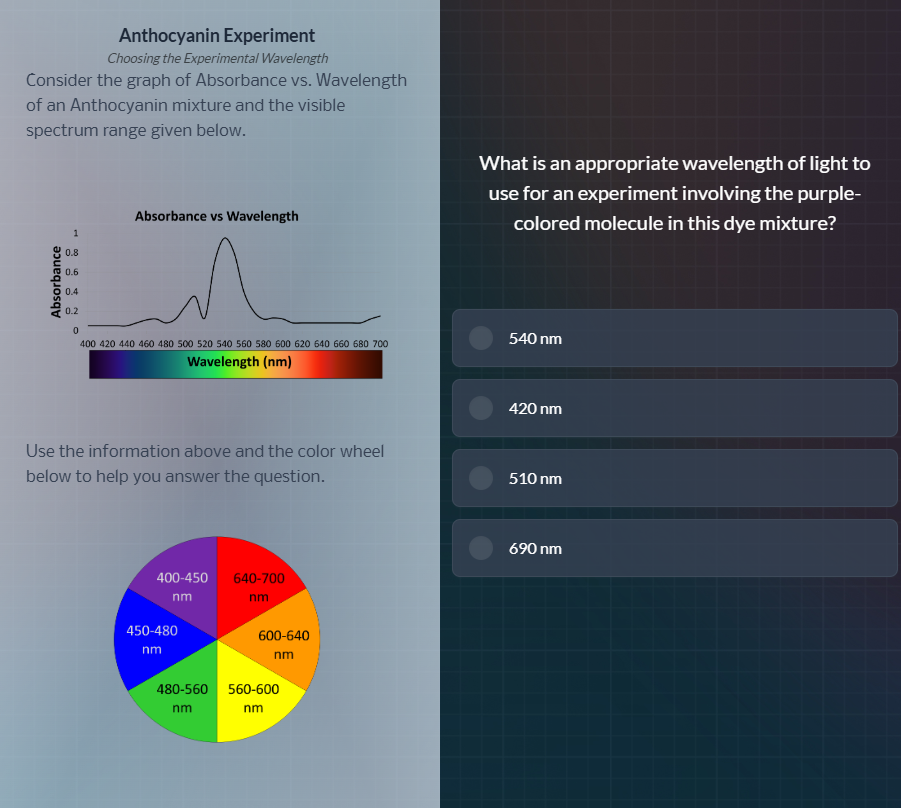
consider the graph of absorbance vs. wavelength of an anthocyanin mixture and the visible spectrum range given below.
absorbance vs wavelength
wavelength (nm)
use the information above and the color wheel below to help you answer the question.
what is an appropriate wavelength of light to use for an experiment involving the purple-colored molecule in this dye mixture?
540 nm
420 nm
510 nm
690 nm
Brief Explanations
- Purple-colored molecules absorb light in the complementary wavelength range. From the color wheel, purple (400-450 nm) complements yellow-green (560-600 nm).
- The absorbance vs. wavelength graph shows the highest absorbance peak occurs around 540-560 nm, which is closest to the complementary range for purple.
- Among the options, 540 nm is the wavelength that aligns with the molecule's strongest absorbance, which is optimal for experimental measurements.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
540 nm