QUESTION IMAGE
Question
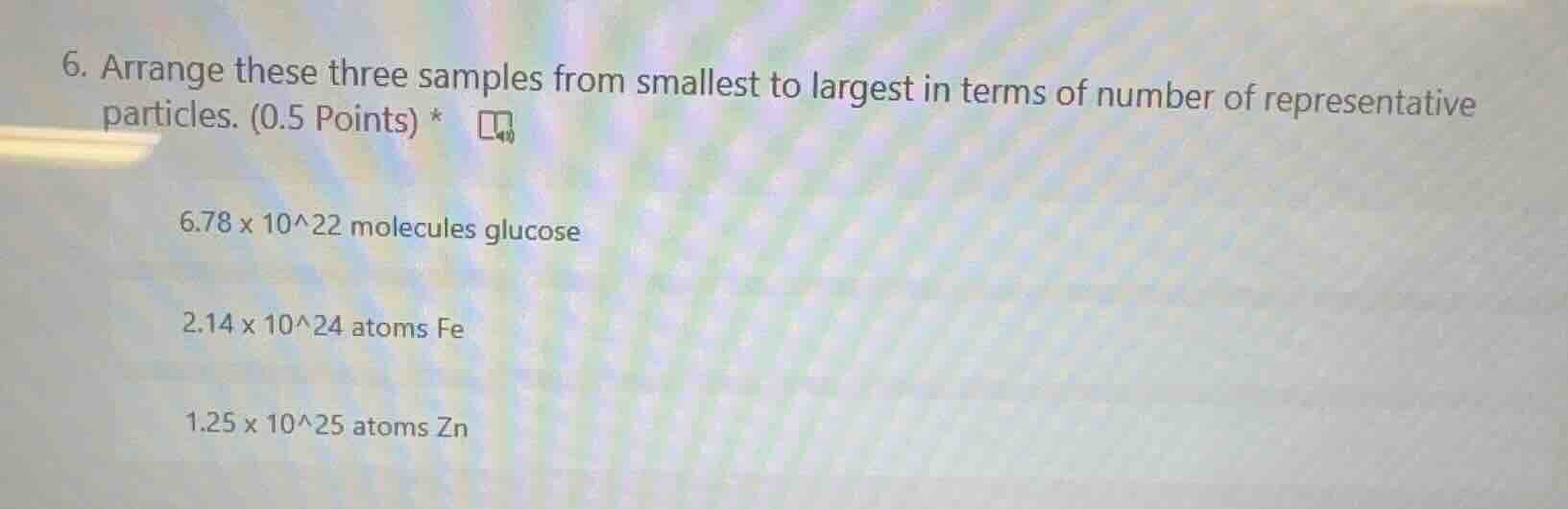
- arrange these three samples from smallest to largest in terms of number of representative particles. (0.5 points) *
6.78 x 10^22 molecules glucose
2.14 x 10^24 atoms fe
1.25 x 10^25 atoms zn
Step1: Analyze the exponents of the numbers
The three samples have the following number of particles:
- Glucose: \(6.78 \times 10^{22}\) molecules
- Fe: \(2.14 \times 10^{24}\) atoms
- Zn: \(1.25 \times 10^{25}\) atoms
We can rewrite the numbers to have the same exponent for easier comparison. Let's convert them to have an exponent of 22.
Step2: Convert Fe's number of particles
For Fe: \(2.14 \times 10^{24}=2.14\times10^{2}\times 10^{22}=214\times 10^{22}\)
Step3: Convert Zn's number of particles
For Zn: \(1.25 \times 10^{25}=1.25\times10^{3}\times 10^{22}=1250\times 10^{22}\)
Step4: Compare the coefficients
Now we have:
- Glucose: \(6.78 \times 10^{22}\)
- Fe: \(214\times 10^{22}\)
- Zn: \(1250\times 10^{22}\)
Since \(6.78<214<1250\), the order from smallest to largest is based on the number of particles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(6.78 \times 10^{22}\) molecules glucose, \(2.14 \times 10^{24}\) atoms Fe, \(1.25 \times 10^{25}\) atoms Zn