QUESTION IMAGE
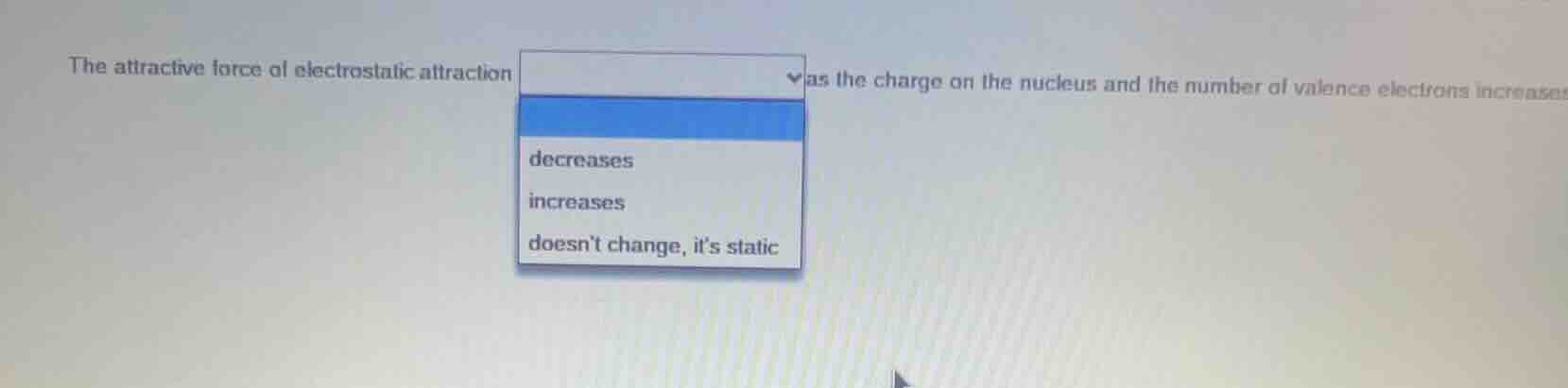
Question
the attractive force of electrostatic attraction as the charge on the nucleus and the number of valence electrons increases decreases increases doesnt change, its static
Brief Explanations
Electrostatic attractive force between a nucleus and valence electrons depends on the magnitudes of the charges involved. Coulomb's law states that force is directly proportional to the product of the two charges. As nuclear charge (positive) and valence electron count (negative charge magnitude) increase, the attractive force grows.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
increases