QUESTION IMAGE
Question
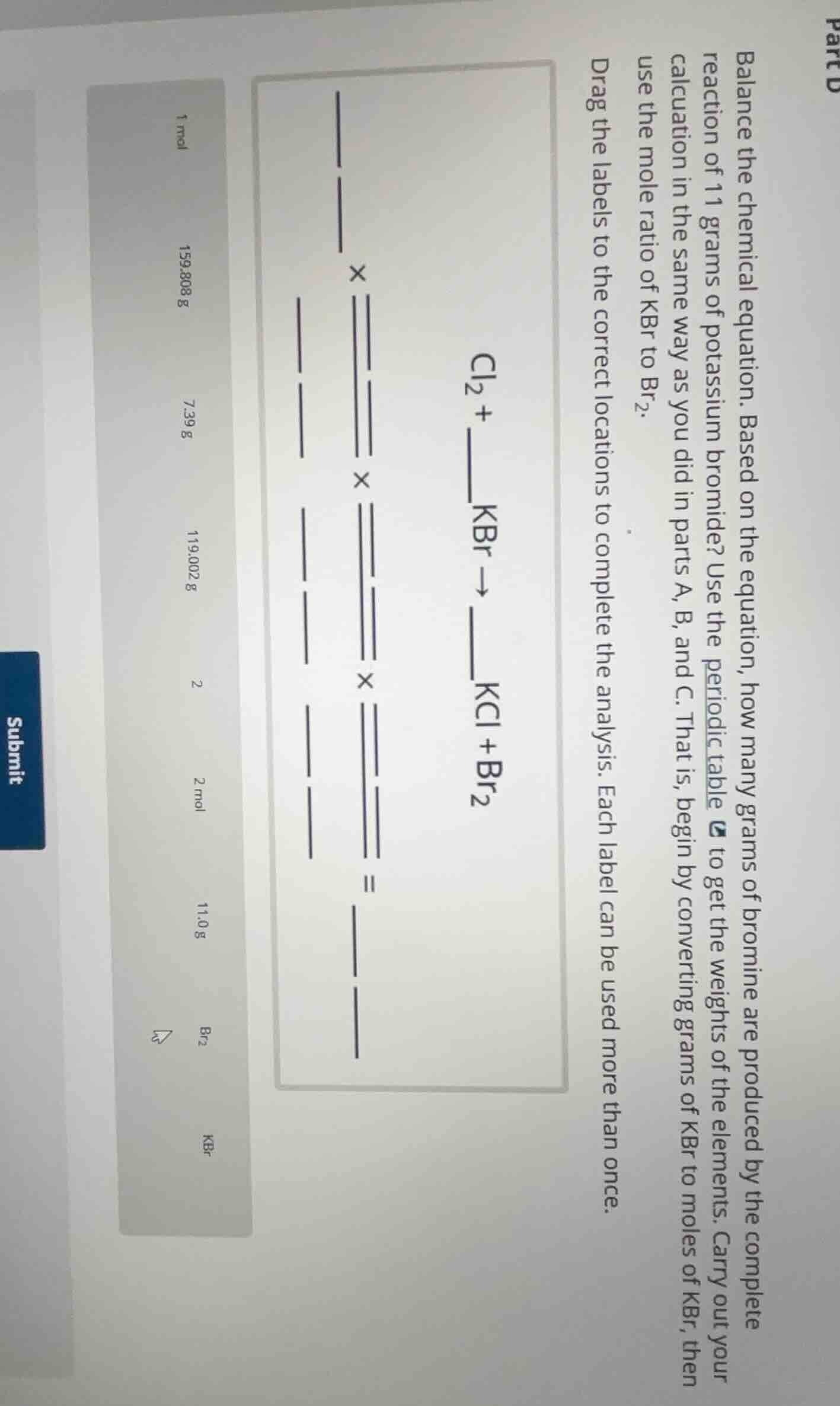
balance the chemical equation. based on the equation, how many grams of bromine are produced by the complete reaction of 11 grams of potassium bromide? use the periodic table to get the weights of the elements. carry out your calculation in the same way as you did in parts a, b, and c. that is, begin by converting grams of kbr to moles of kbr, then use the mole ratio of kbr to br₂. drag the labels to the correct locations to complete the analysis. each label can be used more than once. \\(\ce{cl2 + ___ kbr -> ___ kcl + br2}\\)
Step1: Balance the chemical equation
First, balance the atoms of each element. For the reaction $\text{Cl}_2 + \text{KBr}
ightarrow \text{KCl} + \text{Br}_2$:
- Balance Br: add coefficient 2 to KBr
- Balance K and Cl: add coefficient 2 to KCl
Balanced equation: $\text{Cl}_2 + 2\text{KBr}
ightarrow 2\text{KCl} + \text{Br}_2$
Mole ratio of $\text{KBr}:\text{Br}_2 = 2:1$
Step2: Calculate molar mass of KBr
Molar mass of K ($39.098\ \text{g/mol}$) + molar mass of Br ($79.904\ \text{g/mol}$)
$\text{Molar mass of KBr} = 39.098 + 79.904 = 119.002\ \text{g/mol}$
Step3: Convert mass of KBr to moles
Use mass and molar mass to find moles:
$\text{Moles of KBr} = \frac{\text{Mass of KBr}}{\text{Molar mass of KBr}} = \frac{11.0\ \text{g}}{119.002\ \text{g/mol}}$
Step4: Convert moles of KBr to moles of Br₂
Use mole ratio $2\ \text{mol KBr} : 1\ \text{mol Br}_2$:
$\text{Moles of Br}_2 = \frac{11.0\ \text{g}}{119.002\ \text{g/mol}} \times \frac{1\ \text{mol Br}_2}{2\ \text{mol KBr}}$
Step5: Convert moles of Br₂ to grams
Molar mass of $\text{Br}_2 = 2 \times 79.904 = 159.808\ \text{g/mol}$
$\text{Mass of Br}_2 = \frac{11.0\ \text{g}}{119.002\ \text{g/mol}} \times \frac{1\ \text{mol Br}_2}{2\ \text{mol KBr}} \times 159.808\ \text{g/mol}$
Step6: Calculate final mass
$\text{Mass of Br}_2 = \frac{11.0 \times 159.808}{119.002 \times 2} \approx 7.39\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Balanced equation: $\boldsymbol{\text{Cl}_2 + 2\text{KBr}
ightarrow 2\text{KCl} + \text{Br}_2}$
Mass calculation sequence:
$\boldsymbol{11.0\ \text{g KBr}} \times \boldsymbol{\frac{1\ \text{mol KBr}}{119.002\ \text{g KBr}}} \times \boldsymbol{\frac{1\ \text{mol Br}_2}{2\ \text{mol KBr}}} \times \boldsymbol{159.808\ \text{g Br}_2/\text{mol Br}_2} = \boldsymbol{7.39\ \text{g Br}_2}$
Final mass of bromine produced: $\boldsymbol{7.39\ \text{g}}$