QUESTION IMAGE
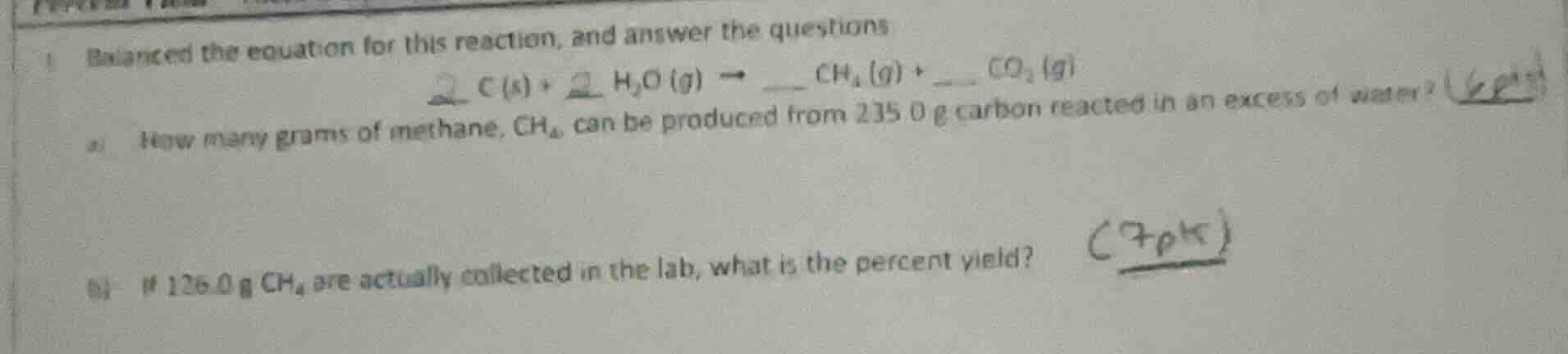
Question
- balance the equation for this reaction, and answer the questions
$\boldsymbol{2 c(s) + 2 h_2o(g)
ightarrow __ ch_4(g) + __ co_2(g)}$
a) how many grams of methane, $ch_4$, can be produced from 235.0 g carbon reacted in an excess of water?
b) if 126.0 g $ch_4$ are actually collected in the lab, what is the percent yield?
Step1: Balance the chemical equation
First, balance the given reaction:
(Verify: C: 2 on left, 1+1=2 on right; H: 4 on left, 4 on right; O: 2 on left, 2 on right)
Step2: Calculate moles of C
Molar mass of C = $12.01\ \text{g/mol}$. Moles of C:
$$n(\text{C}) = \frac{235.0\ \text{g}}{12.01\ \text{g/mol}} \approx 19.57\ \text{mol}$$
Step3: Relate moles of C to CH₄
From balanced equation, 2 mol C produces 1 mol CH₄. Moles of CH₄:
$$n(\text{CH}_4) = 19.57\ \text{mol} \times \frac{1\ \text{mol CH}_4}{2\ \text{mol C}} = 9.785\ \text{mol}$$
Step4: Calculate theoretical mass of CH₄
Molar mass of CH₄ = $12.01 + 4\times1.008 = 16.042\ \text{g/mol}$. Theoretical mass:
$$m_{\text{theoretical}}(\text{CH}_4) = 9.785\ \text{mol} \times 16.042\ \text{g/mol} \approx 157.0\ \text{g}$$
Step5: Calculate percent yield
Percent yield formula: $\text{Percent Yield} = \frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100\%$
$$\text{Percent Yield} = \frac{126.0\ \text{g}}{157.0\ \text{g}} \times 100\% \approx 80.25\%$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) $157.0\ \text{g}$
b) $80.25\%$