QUESTION IMAGE
Question
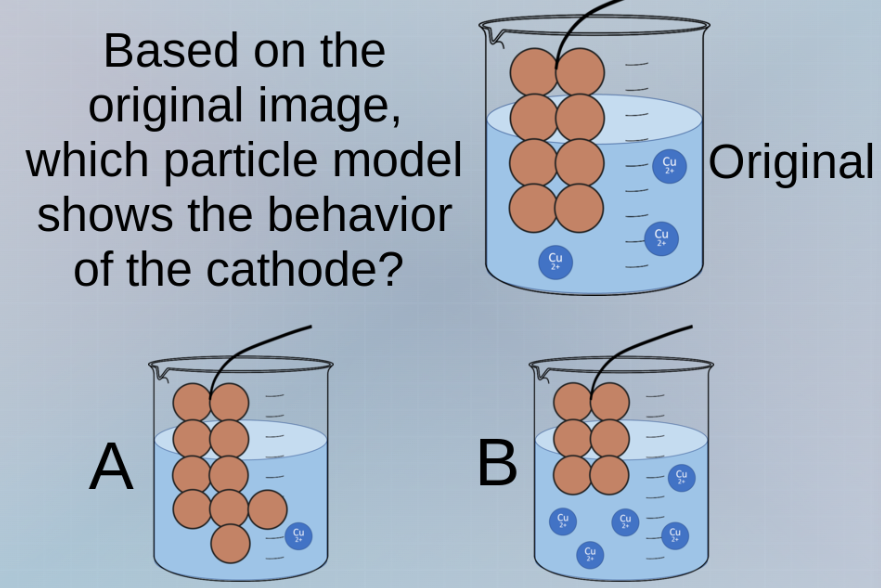
based on the original image, which particle model shows the behavior of the cathode? original a b
In electrolysis, at the cathode (negative electrode), cations (like $\text{Cu}^{2+}$) gain electrons and deposit as solid metal (Cu atoms). The original image has $\text{Cu}^{2+}$ ions in solution and Cu atoms on the electrode. For the cathode behavior, $\text{Cu}^{2+}$ ions should reduce to Cu, increasing the number of Cu atoms (solid) and decreasing $\text{Cu}^{2+}$ ions? Wait, no—wait, if it's a copper plating or electrolysis of $\text{CuSO}_4$ with copper electrodes, at cathode: $\text{Cu}^{2+} + 2e^-
ightarrow \text{Cu}$. So the solid Cu (the spheres) should increase, and the $\text{Cu}^{2+}$ (blue circles) might decrease? Wait, looking at the options:
Original: has some Cu atoms (brown spheres) on the electrode, and $\text{Cu}^{2+}$ in solution.
Option A: More brown spheres (Cu atoms) in the solution? No, Cu is solid, so it should deposit on the electrode, not go into solution. Wait, maybe the electrode is the brown spheres, and at cathode, $\text{Cu}^{2+}$ is reduced to Cu, so the number of brown spheres (Cu) on the electrode increases, and $\text{Cu}^{2+}$ in solution decreases? Wait, no—wait, maybe the setup is electroplating, where the cathode is where Cu²⁺ is reduced to Cu, so the solid Cu (the electrode) gains more atoms, and the $\text{Cu}^{2+}$ concentration in solution decreases? Wait, looking at the images:
Original: electrode has 8 brown spheres (4 rows: 2,2,2,2? Wait, original: top two, then two, two, two—total 8? Wait, original: 2 + 2 + 2 + 2 = 8? Then A: electrode has more brown spheres (like 9? 2,2,2,3? And one in solution? No, Cu is solid, so it shouldn't be in solution. Wait, maybe I got it wrong. Alternatively, if the cathode is where reduction occurs, $\text{Cu}^{2+}$ (aq) → Cu (s). So the solid Cu (the electrode) should have more atoms (brown spheres), and the $\text{Cu}^{2+}$ (blue) in solution should decrease? Wait, no—wait, in option A, there's a brown sphere in the solution, which is incorrect (Cu is solid, so it's on the electrode, not in solution). Option B: more $\text{Cu}^{2+}$? No, that would be oxidation. Wait, maybe the original is a copper electrode in $\text{CuSO}_4$ solution, and it's electrolysis with copper electrodes (electroplating or refining). At cathode: $\text{Cu}^{2+} + 2e^-
ightarrow \text{Cu}$ (deposition, so electrode gains Cu atoms). At anode: $\text{Cu}
ightarrow \text{Cu}^{2+} + 2e^-$. But the question is about the cathode. So the cathode should have more Cu atoms (brown spheres) and less $\text{Cu}^{2+}$? Wait, no—wait, the original has some $\text{Cu}^{2+}$ (blue circles) in solution. Option A: the electrode (brown spheres) has an extra sphere in the solution? No, that's wrong. Option B: more $\text{Cu}^{2+}$? No. Wait, maybe I messed up. Wait, the cathode is where reduction happens, so $\text{Cu}^{2+}$ is reduced to Cu, so the solid Cu (the electrode) should increase (more brown spheres), and the $\text{Cu}^{2+}$ in solution should decrease? Wait, original: electrode has 8 brown spheres, solution has 2 $\text{Cu}^{2+}$. Option A: electrode has 9 brown spheres (one extra in the solution? No, that's a solid, so it should be on the electrode. Wait, maybe the brown spheres are the electrode, and in option A, the electrode has grown (more spheres) and one sphere is in the solution (which is a mistake, but maybe the diagram is simplified). Wait, no—Cu is a solid, so it can't be in the solution. So maybe the correct answer is A? Wait, no—wait, in option B, there are more $\text{Cu}^{2+}$ in solution, which would be if oxidation is happening (anode), but the question is about catho…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Option A